In Vivo Safety of Tumor Treating Fields (TTFields) Applied to the Torso
- PMID: 34249709
- PMCID: PMC8264759
- DOI: 10.3389/fonc.2021.670809
In Vivo Safety of Tumor Treating Fields (TTFields) Applied to the Torso
Abstract
Background: Tumor Treating Fields (TTFields) therapy is a non-invasive, loco-regional, anti-mitotic treatment modality that targets rapidly dividing cancerous cells, utilizing low intensity, alternating electric fields at cancer-cell-type specific frequencies. TTFields therapy is approved for the treatment of newly diagnosed and recurrent glioblastoma (GBM) in the US, Europe, Israel, Japan, and China. The favorable safety profile of TTFields in patients with GBM is partially attributed to the low rate of mitotic events in normal, quiescent brain cells. However, specific safety evaluations are warranted at locations with known high rates of cellular proliferation, such as the torso, which is a primary site of several of the most aggressive malignant tumors.
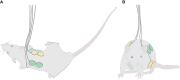
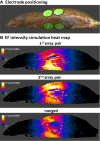
Methods: The safety of delivering TTFields to the torso of healthy rats at 150 or 200 kHz, which were previously identified as optimal frequencies for treating multiple torso cancers, was investigated. Throughout 2 weeks of TTFields application, animals underwent daily clinical examinations, and at treatment cessation blood samples and internal organs were examined. Computer simulations were performed to verify that the targeted internal organs of the torso were receiving TTFields at therapeutic intensities (≥ 1 V/cm root mean square, RMS).
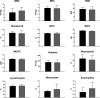
Results: No treatment-related mortality was observed. Furthermore, no significant differences were observed between the TTFields-treated and control animals for all examined safety parameters: activity level, food and water intake, stools, motor neurological status, respiration, weight, complete blood count, blood biochemistry, and pathological findings of internal organs. TTFields intensities of 1 to 2.5 V/cm RMS were confirmed for internal organs within the target region.
Conclusions: This research demonstrates the safety of therapeutic level TTFields at frequencies of 150 and 200 kHz when applied as monotherapy to the torso of healthy rats.
Keywords: Tumor Treating Fields (TTFields); cancer treatment; electric field simulations; safety; torso.
Copyright © 2021 Blatt, Davidi, Munster, Shteingauz, Cahal, Zeidan, Marciano, Bomzon, Haber, Giladi, Weinberg, Kinzel and Palti.
Conflict of interest statement
Authors RB, SD, MM, AS, SC, AZ, TM, ZB, AH, MG, UW, and YP were employed by company Novocure Ltd. Author AK was employed by company Novocure GmbH.
Figures



References
-
- Stupp R, Taillibert S, Kanner A, Read W, Steinberg DM, Lhermitte B, et al. Effect of Tumor-Treating Fields Plus Maintenance Temozolomide vs Maintenance Temozolomide Alone on Survival in Patients With Glioblastoma: A Randomized Clinical Trial. JAMA (2017) 318(23):2306–16. 10.1001/jama.2017.18718 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

