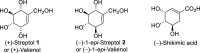
Novel Stereoselective Syntheses of (+)-Streptol and (-)-1 -epi-Streptol Starting from Naturally Abundant (-)-Shikimic Acid
- PMID: 34250367
- PMCID: PMC8264934
- DOI: 10.1021/acsomega.1c02502
Novel Stereoselective Syntheses of (+)-Streptol and (-)-1 -epi-Streptol Starting from Naturally Abundant (-)-Shikimic Acid
Abstract
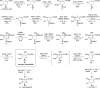
Novel highly stereoselective syntheses of (+)-streptol and (-)-1-epi-streptol starting from naturally abundant (-)-shikimic acid were described in this article. (-)-Shikimic acid was first converted to the common key intermediate by 11 steps in 40% yield. It was then converted to (+)-streptol by three steps in 72% yield, and it was also converted to (-)-1-epi-streptol by one step in 90% yield. In summary, (+)-streptol and (-)-1-epi-streptol were synthesized from (-)-shikimic acid by 14 and 12 steps in 29 and 36% overall yields, respectively.
© 2021 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
References
-
- Shui F.; Jia J.; Yang X.; Zhou Q.; Jiang Y.; Chen X. Synthesis of (+)-epoxydon, (−)-phyllostine, (−)-RKTS 33, and (−)-parasitenone featuring selective sulfonylation and oxirane ring closure of Aldol cyclization products. Eur. J. Org. Chem. 2020, 3981–3988. 10.1002/ejoc.202000486. - DOI
- Osborn A. R.; Kean K. M.; Alseud K. M.; Almabruk K. H.; Asamiza S.; Lee J. A.; Karplus P. A.; Mahmus T. Evolution and distribution of C7–cyclitol synthases in prokaryotes and eukaryotes. ACS Chem. Biol. 2017, 12, 979–988. 10.1021/acschembio.7b00066. - DOI - PMC - PubMed
- Mondal S.; Sureshan K. M. Carbasugar synthesis via vinylogous ketal: total syntheses of (+)-MK7607, (−)-MK7607, (−)-Gabosine A, (−)-Epoxydine B, (−)-Epoxydine C, epi-(+)-Gabosine E and epi-(+)-MK7607. J. Org. Chem. 2016, 81, 11635–11645. 10.1021/acs.joc.6b01876. - DOI - PubMed
- Trapero A.; Egido-Gabas M.; Bujons J.; Llebaria A. Synthesis and evaluation of hydroxymethylaminocyclitols as glycosidase inhibitors. J. Org. Chem. 2015, 80, 3512–3529. 10.1021/acs.joc.5b00133. - DOI - PubMed
- Vidyasagar A.; Sureshan K. M. Total syntheses of five uvacalols: structural validation of uvacalol A, uvacalol B and uvacalol C and disproval of the structures of uvacalol E and uvacalol G. Org. Biomol. Chem. 2015, 13, 3900–3910. 10.1039/C4OB02663B. - DOI - PubMed
- Usami Y.; Mizuki K. Stereostructure reassignment and determination of the absolute configuration of pericosine Do by a synthetic approach. J. Nat. Prod. 2011, 74, 877–881. 10.1021/np100843j. - DOI - PubMed
- Choi W. S.; Xiumei W. X.; Choeng Y. H.; Mahmud T.; Jeong B. C.; Lee S. H.; Chang Y. K.; Kim C-J.; Soon-Kwang Hong S.-K. Genetic organization of the putative salbostatin biosynthetic gene cluster including the 2-epi-5-epi-valiolone synthase gene in Streptomyces albus ATCC 21838. Appl. Microbiol. Biotechnol. 2008, 80, 637–645. 10.1007/s00253-008-1591-2. - DOI - PubMed
- Delgado A. Recent advances in the chemistry of aminocyclitols. Eur. J. Org. Chem. 2008, 3893–3906. 10.1002/ejoc.200800238. - DOI
- Arjona O.; Gómez A. M.; López J. C.; Plumet J. Synthesis and conformational and biological aspects of carbasugars. Chem. Rev. 2007, 107, 1919–2036. 10.1021/cr0203701. - DOI - PubMed
- Mahmud T. The C7N aminocyclitol family of natural products. Nat. Prod. Rep. 2003, 20, 137–166. 10.1039/b205561a. - DOI - PubMed
- Naganawa H.; Hashizume H.; Kubota Y.; Sawa R.; Takahashi Y.; et al. Biosynthesis of the cyclitol moiety of pyralomicin 1a in Nonomuraea spiralis MI178-34F18. J. Antibiot. 2002, 55, 578–584. 10.7164/antibiotics.55.578. - DOI - PubMed
- Marco-Contelles J. Cyclohexane epoxides −chemistry and biochemistry of (+)-cyclophellitol. Eur. J. Org. Chem. 2001, 1607–1618. 10.1002/1099-0690(200105)2001:9<1607::AID-EJOC1607>3.0.CO;2-6. - DOI
-
- Isogai A.; Sakuda S.; Nakayama J.; Watanabe S.; Suzuki A. Isolation and structural elucidation of a new cyclitol derivative, streptol, as a plant growth regulator. Agric. Biol. Chem. 1987, 51, 2277–2279. 10.1080/00021369.1987.10868342. - DOI
- Kizuka M.; Enokita R.; Shibata K.; Okamoto Y.; Inoue Y.; Okazaki T. Studies on actinomycetes isolated from plant leaves. New plant growth inhibitors A-79197-2 and -3 from Dacthylosporangium aurantiacum SANK 61299. Actinomycetologica 2002, 16, 14–16. 10.3209/saj.16_14. - DOI
- Sedmera P.; Halada P.; Pospísil S. New carbasugars from Streptomyces lincolnensis. Magn. Reson. Chem. 2009, 47, 519–522. 10.1002/mrc.2408. - DOI - PubMed
-
- Hsiao C.-C.; Sieber S.; Georgiou A.; Bailly A.; Emmanouilidou D.; Carlier A.; Eberl L.; Gademann K. Synthesis and biological evaluation of the novel growth inhibitor streptol glucoside, isolated from an obligate plant symbiont. Chem. – Eur. J. 2019, 25, 1722–1726. 10.1002/chem.201805693. - DOI - PubMed
- Iwu M.; Okunji C.; Tchimene M.; Anele N.; Chah K.; Osonwa U.; Akpa P. A.; Onunkwo G. C. Stability of cough linctus (streptol) formulated from named medicinal plant extracts. Chem. Pharm. Bull. 2009, 57, 229–232. 10.1248/cpb.57.229. - DOI - PubMed
-
- Zhang C.-S.; Stratmann A.; Block O.; Bruckner R.; Podeschwa M.; Altenbach H.-J.; Wehmeier U. F.; Piepersberg W. Biosynthesis of the C7-cyclitol moiety of acarbose in Actinoplanes species SE50/110. 7-O-Phosphorylation of the initial cyclitol precursor leads to proposal of a new biosynthetic pathway. J. Biol. Chem. 2002, 277, 22853–22862. 10.1074/jbc.M202375200. - DOI - PubMed
- Wehmeier U. F. The biosynthesis and metabolism of acarbose in Actinoplanes sp. SE 50/110: A progress report. Biocatal. Biotransform. 2003, 21, 279–284. 10.1080/10242420310001614388. - DOI
- Zhang C.-S.; Podeschwa M.; Block O.; Altenbach H.-J.; Piepersberg W.; Wehmeier U. F. Identification of a 1-epi-valienol 7-kinase activity in the producer of acarbose, Actinoplanes sp. SE50/110. FEBS Lett. 2003, 540, 53–57. 10.1016/S0014-5793(03)00222-9. - DOI - PubMed
LinkOut - more resources
Full Text Sources