Snake extract-laden hemostatic bioadhesive gel cross-linked by visible light
- PMID: 34261653
- PMCID: PMC8279511
- DOI: 10.1126/sciadv.abf9635
Snake extract-laden hemostatic bioadhesive gel cross-linked by visible light
Abstract
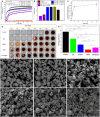
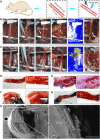
Bioadhesives reduce operation time and surgical complications. However, in the presence of blood, adhesion strength is often compromised. Inspired by the blood clotting activity of snake venom, we report a visible light-induced blood-resistant hemostatic adhesive (HAD) containing gelatin methacryloyl and reptilase, which is a hemocoagulase (HC) extracted from Bothrops atrox HAD leads to the activation and aggregation of platelets and efficiently transforms fibrinogen into fibrin to achieve rapid hemostasis and seal the tissue. Blood clotting time with HAD was about 45 s compared with 5 to 6 min without HAD. HAD instantaneously achieved hemostasis on liver incision (~45 s) and cut rat tail (~34 s) and reduced blood loss by 79 and 78%, respectively. HAD is also efficient in sealing severely injured liver and abdominal aorta. HAD has great potential to bridge injured tissues by combing hemostasis with adhesives.
Copyright © 2021 The Authors, some rights reserved; exclusive licensee American Association for the Advancement of Science. No claim to original U.S. Government Works. Distributed under a Creative Commons Attribution NonCommercial License 4.0 (CC BY-NC).
Figures








References
-
- Yuk H., Varela C. E., Nabzdyk C. S., Mao X., Padera R. F., Roche E. T., Zhao X., Dry double-sided tape for adhesion of wet tissues and devices. Nature 575, 169–174 (2019). - PubMed
-
- Xu X., Xia X., Zhang K., Rai A., Li Z., Zhao P., Wei K., Zou L., Yang B., Wong W.-K., Chiu P. W.-Y., Bian L., Bioadhesive hydrogels demonstrating pH-independent and ultrafast gelation promote gastric ulcer healing in pigs. Sci. Transl. Med. 12, eaba8014 (2020). - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

