Impaired respiratory burst contributes to infections in PKCδ-deficient patients
- PMID: 34264265
- PMCID: PMC8288504
- DOI: 10.1084/jem.20210501
Impaired respiratory burst contributes to infections in PKCδ-deficient patients
Abstract
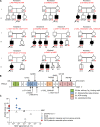
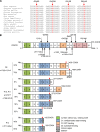
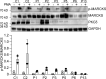
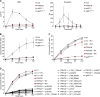
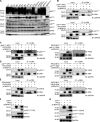
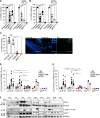
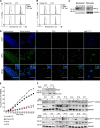
Patients with autosomal recessive protein kinase C δ (PKCδ) deficiency suffer from childhood-onset autoimmunity, including systemic lupus erythematosus. They also suffer from recurrent infections that overlap with those seen in patients with chronic granulomatous disease (CGD), a disease caused by defects of the phagocyte NADPH oxidase and a lack of reactive oxygen species (ROS) production. We studied an international cohort of 17 PKCδ-deficient patients and found that their EBV-B cells and monocyte-derived phagocytes produced only small amounts of ROS and did not phosphorylate p40phox normally after PMA or opsonized Staphylococcus aureus stimulation. Moreover, the patients' circulating phagocytes displayed abnormally low levels of ROS production and markedly reduced neutrophil extracellular trap formation, altogether suggesting a role for PKCδ in activation of the NADPH oxidase complex. Our findings thus show that patients with PKCδ deficiency have impaired NADPH oxidase activity in various myeloid subsets, which may contribute to their CGD-like infectious phenotype.
© 2021 Neehus et al.
Conflict of interest statement
Disclosures: P.A. Brogan reported personal fees from Sobi, Novartis, Roche, and GSK, and grants from Sobi outside the submitted work. No other disclosures were reported.
Figures






References
-
- Belot, A., Kasher P.R., Trotter E.W., Foray A.-P., Debaud A.-L., Rice G.I., Szynkiewicz M., Zabot M.-T., Rouvet I., Bhaskar S.S., et al. 2013. Protein kinase cδ deficiency causes mendelian systemic lupus erythematosus with B cell-defective apoptosis and hyperproliferation. Arthritis Rheum. 65:2161–2171. 10.1002/art.38008 - DOI - PMC - PubMed
-
- Bey, E.A., Xu B., Bhattacharjee A., Oldfield C.M., Zhao X., Li Q., Subbulakshmi V., Feldman G.M., Wientjes F.B., and Cathcart M.K.. 2004. Protein kinase C δ is required for p47phox phosphorylation and translocation in activated human monocytes. J. Immunol. 173:5730–5738. 10.4049/jimmunol.173.9.5730 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- DH_/Department of Health/United Kingdom
- 8UL1TR001866/NH/NIH HHS/United States
- R37 AI095983/AI/NIAID NIH HHS/United States
- R01 AI127564/AI/NIAID NIH HHS/United States
- RR/NCRR NIH HHS/United States
- UM1 HG006504/HG/NHGRI NIH HHS/United States
- UM1HG006504/HG/NHGRI NIH HHS/United States
- UL1 TR001863/TR/NCATS NIH HHS/United States
- R01 AI095983/AI/NIAID NIH HHS/United States
- HHMI/Howard Hughes Medical Institute/United States
- UL1 TR001866/TR/NCATS NIH HHS/United States
- R01 AI089970/AI/NIAID NIH HHS/United States
- 5R01AI089970/AI/NIAID NIH HHS/United States
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

