Identification of Compounds for Butyrylcholinesterase Inhibition
- PMID: 34269114
- PMCID: PMC8637366
- DOI: 10.1177/24725552211030897
Identification of Compounds for Butyrylcholinesterase Inhibition
Abstract
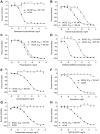
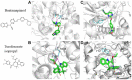
Butyrylcholinesterase (BChE) is a nonspecific cholinesterase enzyme that hydrolyzes choline-based esters. BChE plays a critical role in maintaining normal cholinergic function like acetylcholinesterase (AChE) through hydrolyzing acetylcholine (ACh). Selective BChE inhibition has been regarded as a viable therapeutic approach in Alzheimer's disease. As of now, a limited number of selective BChE inhibitors are available. To identify BChE inhibitors rapidly and efficiently, we have screened 8998 compounds from several annotated libraries against an enzyme-based BChE inhibition assay in a quantitative high-throughput screening (qHTS) format. From the primary screening, we identified a group of 125 compounds that were further confirmed to inhibit BChE activity, including previously reported BChE inhibitors (e.g., bambuterol and rivastigmine) and potential novel BChE inhibitors (e.g., pancuronium bromide and NNC 756), representing diverse structural classes. These BChE inhibitors were also tested for their selectivity by comparing their IC50 values in BChE and AChE inhibition assays. The binding modes of these compounds were further studied using molecular docking analyses to identify the differences between the interactions of these BChE inhibitors within the active sites of AChE and BChE. Our qHTS approach allowed us to establish a robust and reliable process to screen large compound collections for potential BChE inhibitors.
Keywords: BChE inhibitors; butyrylcholinesterase; colorimetric BChE assay; molecular docking; qHTS.
Conflict of interest statement
Figures

Similar articles
-
Identification of Potent and Selective Acetylcholinesterase/Butyrylcholinesterase Inhibitors by Virtual Screening.J Chem Inf Model. 2023 Apr 24;63(8):2321-2330. doi: 10.1021/acs.jcim.3c00230. Epub 2023 Apr 3. J Chem Inf Model. 2023. PMID: 37011147 Free PMC article.
-
Design, synthesis, and evaluation of novel cinnamic acid-tryptamine hybrid for inhibition of acetylcholinesterase and butyrylcholinesterase.Daru. 2020 Dec;28(2):463-477. doi: 10.1007/s40199-020-00346-9. Epub 2020 May 5. Daru. 2020. PMID: 32372339 Free PMC article.
-
Rivastigmine-Bambuterol Hybrids as Selective Butyrylcholinesterase Inhibitors.Molecules. 2023 Dec 22;29(1):72. doi: 10.3390/molecules29010072. Molecules. 2023. PMID: 38202655 Free PMC article.
-
Butyrylcholinesterase Protein Ends in the Pathogenesis of Alzheimer's Disease-Could BCHE Genotyping Be Helpful in Alzheimer's Therapy?Biomolecules. 2019 Oct 9;9(10):592. doi: 10.3390/biom9100592. Biomolecules. 2019. PMID: 31601022 Free PMC article. Review.
-
Cholinesterase Inhibitory Activity of Some semi-Rigid Spiro Heterocycles: POM Analyses and Crystalline Structure of Pharmacophore Site.Mini Rev Med Chem. 2018;18(8):711-716. doi: 10.2174/1389557517666170713114039. Mini Rev Med Chem. 2018. PMID: 28714400 Review.
Cited by
-
Design, Synthesis and Biological Evaluation of Biscarbamates as Potential Selective Butyrylcholinesterase Inhibitors for the Treatment of Alzheimer's Disease.Pharmaceuticals (Basel). 2022 Sep 30;15(10):1220. doi: 10.3390/ph15101220. Pharmaceuticals (Basel). 2022. PMID: 36297332 Free PMC article.
-
Umbelliferone and Its Synthetic Derivatives as Suitable Molecules for the Development of Agents with Biological Activities: A Review of Their Pharmacological and Therapeutic Potential.Pharmaceuticals (Basel). 2023 Dec 15;16(12):1732. doi: 10.3390/ph16121732. Pharmaceuticals (Basel). 2023. PMID: 38139858 Free PMC article. Review.
-
Identification of Potent and Selective Acetylcholinesterase/Butyrylcholinesterase Inhibitors by Virtual Screening.J Chem Inf Model. 2023 Apr 24;63(8):2321-2330. doi: 10.1021/acs.jcim.3c00230. Epub 2023 Apr 3. J Chem Inf Model. 2023. PMID: 37011147 Free PMC article.
-
Synthesis, biological evaluation, molecular docking, and MD simulation of novel 2,4-disubstituted quinazoline derivatives as selective butyrylcholinesterase inhibitors and antioxidant agents.Sci Rep. 2024 Jul 6;14(1):15577. doi: 10.1038/s41598-024-66424-z. Sci Rep. 2024. PMID: 38971857 Free PMC article.
-
Cholinesterase Inhibitors from Plants and Their Potential in Alzheimer's Treatment: Systematic Review.Brain Sci. 2025 Feb 19;15(2):215. doi: 10.3390/brainsci15020215. Brain Sci. 2025. PMID: 40002547 Free PMC article. Review.
References
-
- Taylor P., Radic Z. The Cholinesterases—From Genes to Proteins. Annu. Rev. Pharmacol. 1994, 34, 281–320. - PubMed
-
- Li B., Sedlacek M., Manoharan I., et al.. Butyrylcholinesterase, Paraoxonase, and Albumin Esterase, but Not Carboxylesterase, Are Present in Human Plasma. Biochem. Pharmacol. 2005, 70, 1673–1684. - PubMed
-
- Lockridge O. Genetic Variants of Human Serum Cholinesterase Influence Metabolism of the Muscle Relaxant Succinylcholine. Pharmacol. Ther. 1990, 47, 35–60. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

