Localization, proteomics, and metabolite profiling reveal a putative vesicular transporter for UDP-glucose
- PMID: 34269178
- PMCID: PMC8373376
- DOI: 10.7554/eLife.65417
Localization, proteomics, and metabolite profiling reveal a putative vesicular transporter for UDP-glucose
Abstract
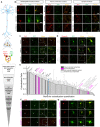
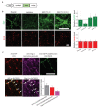
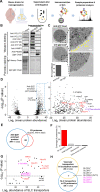
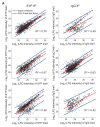
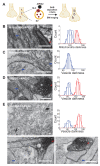
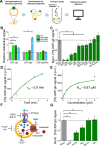
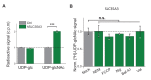
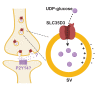
Vesicular neurotransmitter transporters (VNTs) mediate the selective uptake and enrichment of small-molecule neurotransmitters into synaptic vesicles (SVs) and are therefore a major determinant of the synaptic output of specific neurons. To identify novel VNTs expressed on SVs (thus identifying new neurotransmitters and/or neuromodulators), we conducted localization profiling of 361 solute carrier (SLC) transporters tagging with a fluorescent protein in neurons, which revealed 40 possible candidates through comparison with a known SV marker. We parallelly performed proteomics analysis of immunoisolated SVs and identified seven transporters in overlap. Ultrastructural analysis further supported that one of the transporters, SLC35D3, localized to SVs. Finally, by combining metabolite profiling with a radiolabeled substrate transport assay, we identified UDP-glucose as the principal substrate for SLC35D3. These results provide new insights into the functional role of SLC transporters in neurotransmission and improve our understanding of the molecular diversity of chemical transmitters.
Keywords: UDP-glucose; human; mouse; neuroscience; neurotransmitters; solute carrier; synaptic vesicles; transporter.
© 2021, Qian et al.
Conflict of interest statement
CQ, ZW, RS, HY, JZ, YR, YL No competing interests declared
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

