Metabolism and biomarkers of heterocyclic aromatic amines in humans
- PMID: 34271992
- PMCID: PMC8284014
- DOI: 10.1186/s41021-021-00200-7
Metabolism and biomarkers of heterocyclic aromatic amines in humans
Abstract
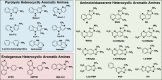
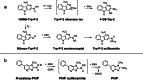
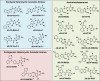
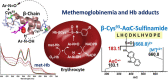
Heterocyclic aromatic amines (HAAs) form during the high-temperature cooking of meats, poultry, and fish. Some HAAs also arise during the combustion of tobacco. HAAs are multisite carcinogens in rodents, inducing cancer of the liver, gastrointestinal tract, pancreas, mammary, and prostate glands. HAAs undergo metabolic activation by N-hydroxylation of the exocyclic amine groups to produce the proposed reactive intermediate, the heteroaryl nitrenium ion, which is the critical metabolite implicated in DNA damage and genotoxicity. Humans efficiently convert HAAs to these reactive intermediates, resulting in HAA protein and DNA adduct formation. Some epidemiologic studies have reported an association between frequent consumption of well-done cooked meats and elevated cancer risk of the colorectum, pancreas, and prostate. However, other studies have reported no associations between cooked meat and these cancer sites. A significant limitation in epidemiology studies assessing the role of HAAs and cooked meat in cancer risk is their reliance on food frequency questionnaires (FFQ) to gauge HAA exposure. FFQs are problematic because of limitations in self-reported dietary history accuracy, and estimating HAA intake formed in cooked meats at the parts-per-billion level is challenging. There is a critical need to establish long-lived biomarkers of HAAs for implementation in molecular epidemiology studies designed to assess the role of HAAs in health risk. This review article highlights the mechanisms of HAA formation, mutagenesis and carcinogenesis, the metabolism of several prominent HAAs, and the impact of critical xenobiotic-metabolizing enzymes on biological effects. The analytical approaches that have successfully biomonitored HAAs and their biomarkers for molecular epidemiology studies are presented.
Keywords: Biomarkers; Cancer; Cooked meat; Heterocyclic aromatic amines; Metabolism; Mutagens.
© 2021. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures











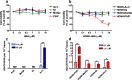
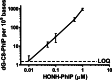


References
-
- Sugimura T, Nagao N, Kawachi T, Honda M, Yahagi T, Seino Y, Stao S, Matsukura N, Matsushima T, Shirai A, Sawamura M, Matsumoto H. Mutagen-carcinogens in food, with special reference to highly mutagenic pyrolytic products in broiled foods. Cold Spring Harbor: Hiatt HH, Watson JD, Winstein JA, Cold Spring Harbor Laboratory; 1977. pp. 1561–1577.
-
- Wakabayashi K, Kim IS, Kurosaka R, Yamaizumi Z, Ushiyama H, Takahashi M, Koyota S, Tada A, Nukaya H, Goto S. Identification of new mutagenic heterocyclic amines and quantification of known heterocyclic amines. Princeton: Adamson RH, Gustafson DR, Ito N, Nagao M, Sugimura T, Wakabayashi K, Yamazoe Y, Princeton Scientific Publishing Co., Inc; 1995. pp. 39–49. - PubMed
-
- Wakabayashi K, Nagao M, Esumi H, Sugimura T. Food-derived mutagens and carcinogens. Cancer Res. 1992;52:2092s–2098s. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

