Altered cellular localisation and expression, together with unconventional protein trafficking, of prion protein, PrPC, in type 1 diabetes
- PMID: 34274990
- PMCID: PMC8715394
- DOI: 10.1007/s00125-021-05501-8
Altered cellular localisation and expression, together with unconventional protein trafficking, of prion protein, PrPC, in type 1 diabetes
Abstract
Aims/hypothesis: Normal cellular prion protein (PrPC) is a conserved mammalian glycoprotein found on the outer plasma membrane leaflet through a glycophosphatidylinositol anchor. Although PrPC is expressed by a wide range of tissues throughout the body, the complete repertoire of its functions has not been fully determined. The misfolded pathogenic isoform PrPSc (the scrapie form of PrP) is a causative agent of neurodegenerative prion diseases. The aim of this study is to evaluate PrPC localisation, expression and trafficking in pancreases from organ donors with and without type 1 diabetes and to infer PrPC function through studies on interacting protein partners.
Methods: In order to evaluate localisation and trafficking of PrPC in the human pancreas, 12 non-diabetic, 12 type 1 diabetic and 12 autoantibody-positive organ donor tissue samples were analysed using immunofluorescence analysis. Furthermore, total RNA was isolated from 29 non-diabetic, 29 type 1 diabetic and 24 autoantibody-positive donors to estimate PrPC expression in the human pancreas. Additionally, we performed PrPC-specific immunoblot analysis on total pancreatic protein from non-diabetic and type 1 diabetic organ donors to test whether changes in PrPC mRNA levels leads to a concomitant increase in PrPC protein levels in human pancreases.
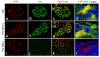
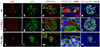
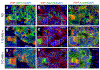
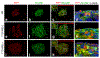
Results: In non-diabetic and type 1 diabetic pancreases (the latter displaying both insulin-positive [INS(+)] and -negative [INS(-)] islets), we found PrPC in islets co-registering with beta cells in all INS(+) islets and, strikingly, unexpected activation of PrPC in alpha cells within diabetic INS(-) islets. We found PrPC localised to the plasma membrane and endoplasmic reticulum (ER) but not the Golgi, defining two cellular pools and an unconventional protein trafficking mechanism bypassing the Golgi. We demonstrate PrPC co-registration with established protein partners, neural cell adhesion molecule 1 (NCAM1) and stress-inducible phosphoprotein 1 (STI1; encoded by STIP1) on the plasma membrane and ER, respectively, linking PrPC function with cyto-protection, signalling, differentiation and morphogenesis. We demonstrate that both PRNP (encoding PrPC) and STIP1 gene expression are dramatically altered in type 1 diabetic and autoantibody-positive pancreases.
Conclusions/interpretation: As the first study to address PrPC expression in non-diabetic and type 1 diabetic human pancreas, we provide new insights for PrPC in the pathogenesis of type 1 diabetes. We evaluated the cell-type specific expression of PrPC in the human pancreas and discovered possible connections with potential interacting proteins that we speculate might address mechanisms relevant to the role of PrPC in the human pancreas.
Keywords: Cellular prion protein; Protein trafficking; Stress-induced phosphoprotein 1; Type 1 diabetes; Type 1 diabetes-dependent endocrine cell expression.
© 2021. The Author(s), under exclusive licence to Springer-Verlag GmbH Germany, part of Springer Nature.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

