Anti-platelet factor 4 antibodies causing VITT do not cross-react with SARS-CoV-2 spike protein
- PMID: 34280256
- PMCID: PMC8294553
- DOI: 10.1182/blood.2021012938
Anti-platelet factor 4 antibodies causing VITT do not cross-react with SARS-CoV-2 spike protein
Abstract
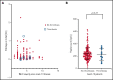
Vaccine-induced immune thrombotic thrombocytopenia (VITT) is a severe adverse effect of ChAdOx1 nCoV-19 COVID-19 vaccine (Vaxzevria) and Janssen Ad26.COV2.S COVID-19 vaccine, and it is associated with unusual thrombosis. VITT is caused by anti-platelet factor 4 (PF4) antibodies activating platelets through their FcγRIIa receptors. Antibodies that activate platelets through FcγRIIa receptors have also been identified in patients with COVID-19. These findings raise concern that vaccination-induced antibodies against anti-SARS-CoV-2 spike protein cause thrombosis by cross-reacting with PF4. Immunogenic epitopes of PF4 and SARS-CoV-2 spike protein were compared using in silico prediction tools and 3D modeling. The SARS-CoV-2 spike protein and PF4 share at least 1 similar epitope. Reactivity of purified anti-PF4 antibodies from patients with VITT was tested against recombinant SARS-CoV-2 spike protein. However, none of the affinity-purified anti-PF4 antibodies from 14 patients with VITT cross-reacted with SARS-CoV-2 spike protein. Sera from 222 polymerase chain reaction-confirmed patients with COVID-19 from 5 European centers were tested by PF4-heparin enzyme-linked immunosorbent assays and PF4-dependent platelet activation assays. We found anti-PF4 antibodies in sera from 19 (8.6%) of 222 patients with COVID-19. However, only 4 showed weak to moderate platelet activation in the presence of PF4, and none of those patients developed thrombotic complications. Among 10 (4.5%) of 222 patients who had COVID-19 with thrombosis, none showed PF4-dependent platelet-activating antibodies. In conclusion, antibodies against PF4 induced by vaccination do not cross-react with the SARS-CoV-2 spike protein, indicating that the intended vaccine-induced immune response against SARS-CoV-2 spike protein is not the trigger of VITT. PF4-reactive antibodies found in patients with COVID-19 in this study were not associated with thrombotic complications.
© 2021 by The American Society of Hematology.
Figures


Comment in
-
COVID-19 and VITT: same or different?Blood. 2021 Oct 7;138(14):1206-1207. doi: 10.1182/blood.2021013362. Blood. 2021. PMID: 34618003 Free PMC article. No abstract available.
References
-
- Paul-Ehrlich-Institut (Bundesinstitut für Impfstoffe und biomedizinische Arzneimittel) . Service-Presse-Sehr seltene Fälle des “Thrombose-mit-Thrombozytopenie Syndroms (TTS) nach Impfung. 2021. https://www.pei.de/DE/service/presse/aktuelles/aktuelles-inhalt.html%20a.... Accessed 31 May 2021.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

