Impact of the introduction of pneumococcal conjugate vaccination on invasive pneumococcal disease and pneumonia in The Gambia: 10 years of population-based surveillance
- PMID: 34280357
- PMCID: PMC8384632
- DOI: 10.1016/S1473-3099(20)30880-X
Impact of the introduction of pneumococcal conjugate vaccination on invasive pneumococcal disease and pneumonia in The Gambia: 10 years of population-based surveillance
Abstract
Background: The Gambia introduced seven-valent pneumococcal conjugate vaccine (PCV7) in August 2009, followed by PCV13 in May, 2011, using a schedule of three primary doses without a booster dose or catch-up immunisation. We aimed to assess the long-term impact of PCV on disease incidence.
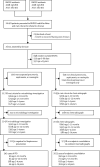
Methods: We did 10 years of population-based surveillance for invasive pneumococcal disease (IPD) and WHO defined radiological pneumonia with consolidation in rural Gambia. The surveillance population included all Basse Health and Demographic Surveillance System residents aged 2 months or older. Nurses screened all outpatients and inpatients at all health facilities using standardised criteria for referral. Clinicians then applied criteria for patient investigation. We defined IPD as a compatible illness with isolation of Streptococcus pneumoniae from a normally sterile site (cerebrospinal fluid, blood, or pleural fluid). We compared disease incidence between baseline (May 12, 2008-May 11, 2010) and post-vaccine years (2016-2017), in children aged 2 months to 14 years, adjusting for changes in case ascertainment over time.
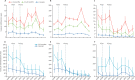
Findings: We identified 22 728 patients for investigation and detected 342 cases of IPD and 2623 cases of radiological pneumonia. Among children aged 2-59 months, IPD incidence declined from 184 cases per 100 000 person-years to 38 cases per 100 000 person-years, an 80% reduction (95% CI 69-87). Non-pneumococcal bacteraemia incidence did not change significantly over time (incidence rate ratio 0·88; 95% CI, 0·64-1·21). We detected zero cases of vaccine-type IPD in the 2-11 month age group in 2016-17. Incidence of radiological pneumonia decreased by 33% (95% CI 24-40), from 10·5 to 7·0 per 1000 person-years in the 2-59 month age group, while pneumonia hospitalisations declined by 27% (95% CI 22-31). In the 5-14 year age group, IPD incidence declined by 69% (95% CI -28 to 91) and radiological pneumonia by 27% (95% CI -5 to 49).
Interpretation: Routine introduction of PCV13 substantially reduced the incidence of childhood IPD and pneumonia in rural Gambia, including elimination of vaccine-type IPD in infants. Other low-income countries can expect substantial impact from the introduction of PCV13 using a schedule of three primary doses.
Funding: Gavi, The Vaccine Alliance; Bill & Melinda Gates Foundation; UK Medical Research Council; Pfizer Ltd.
Copyright © 2021 The Author(s). Published by Elsevier Ltd. This is an Open Access article under the CC BY 4.0 license. Published by Elsevier Ltd.. All rights reserved.
Conflict of interest statement
Declaration of interests RA was employed by GlaxoSmithKline Vaccines and received grant awards from WHO, Gavi, and the Bill & Melinda Gates Foundation whilst employed at MRC Gambia. MK, SH, and BG received grants from the Bill & Melinda Gates Foundation. MK received grants from Gavi, Merck, and Pfizer, and personal fees from Pfizer. All other authors declare no competing interest.
Figures
References
-
- O'Brien KL, Wolfson LJ, Watt JP. Burden of disease caused by Streptococcus pneumoniae in children younger than 5 years: global estimates. Lancet. 2009;374:893–902. - PubMed
-
- von Gottberg A, de Gouveia L, Tempia S. Effects of vaccination on invasive pneumococcal disease in South Africa. N Engl J Med. 2014;371:1889–1899. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

