In-House Filtration Efficiency Assessment of Vapor Hydrogen Peroxide Decontaminated Filtering Facepiece Respirators (FFRs)
- PMID: 34281105
- PMCID: PMC8297238
- DOI: 10.3390/ijerph18137169
In-House Filtration Efficiency Assessment of Vapor Hydrogen Peroxide Decontaminated Filtering Facepiece Respirators (FFRs)
Abstract
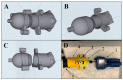
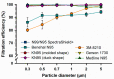
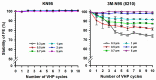
To cope with the shortage of filtering facepiece respirators (FFRs) caused by the coronavirus disease (COVID-19), healthcare institutions have been forced to reuse FFRs using different decontamination methods, including vapor hydrogen peroxide (VHP). However, most healthcare institutions still struggle with evaluating the effect of VHP on filtration efficiency (FE) of the decontaminated FFRs. We developed a low-cost in-house FE assessment using a novel 3D-printed air duct. Furthermore, we assessed the FE of seven types of FFRs. Following 10 VHP cycles, we evaluated the FE of KN95 and 3M-N95 masks. The 3M-N95 and Benehal-N95 masks showed significant lower FE (80.4-91.8%) at fine particle sizes (0.3-1 µm) compared to other FFRs (FE ≥ 98.1%, p < 0.05). Following 10 VHP cycles, the FE of KN95 masks was almost stable (FE stability > 99.1%) for all particle sizes, while 3M-N95 masks were stable only at 2 and 5 µm (FE stability > 98.0%). Statistically, FE stability of 3M-N95 masks at 0.3, 0.5, and 0.7 µm was significantly lower (p ≤ 0.006) than 2 and 5 µm. The in-house FE assessment may be used as an emergency procedure to validate the decontaminated FFRs, as well as a screening option for production control of FFRs. Following VHP cycles, both masks showed high stability at 5 µm, the size of the most suspected droplets implicated in COVID-19 transmission.
Keywords: COVID-19; FFRs; KN95 masks; N95 masks; decontamination; filtration efficiency; vapor hydrogen peroxide.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Effect of gamma irradiation on filtering facepiece respirators and SARS-CoV-2 detection.Sci Rep. 2021 Oct 6;11(1):19888. doi: 10.1038/s41598-021-99414-6. Sci Rep. 2021. PMID: 34615977 Free PMC article.
-
Vapourized hydrogen peroxide decontamination in a hospital setting inactivates SARS-CoV-2 and HCoV-229E without compromising filtration efficiency of unexpired N95 respirators.Am J Infect Control. 2021 Oct;49(10):1227-1231. doi: 10.1016/j.ajic.2021.07.012. Epub 2021 Jul 25. Am J Infect Control. 2021. PMID: 34320409 Free PMC article.
-
Decontamination and reuse of surgical masks and N95 filtering facepiece respirators during the COVID-19 pandemic: A systematic review.Infect Control Hosp Epidemiol. 2021 Jan;42(1):25-30. doi: 10.1017/ice.2020.379. Epub 2020 Jul 30. Infect Control Hosp Epidemiol. 2021. PMID: 32729444 Free PMC article.
-
Exploring options for reprocessing of N95 Filtering Facepiece Respirators (N95-FFRs) amidst COVID-19 pandemic: A systematic review.PLoS One. 2020 Nov 20;15(11):e0242474. doi: 10.1371/journal.pone.0242474. eCollection 2020. PLoS One. 2020. PMID: 33216795 Free PMC article.
-
Filtration performance of three models of N95 filtering facepiece respirators following clinical usage and vaporized hydrogen peroxide decontamination.J Hosp Infect. 2023 Jan;131:122-125. doi: 10.1016/j.jhin.2022.09.026. Epub 2022 Oct 19. J Hosp Infect. 2023. PMID: 36272553
Cited by
-
Effect of gamma irradiation on filtering facepiece respirators and SARS-CoV-2 detection.Sci Rep. 2021 Oct 6;11(1):19888. doi: 10.1038/s41598-021-99414-6. Sci Rep. 2021. PMID: 34615977 Free PMC article.
-
Impact of ultraviolet radiation on filtering facepiece respirators and SARS-CoV-2 detection.Front Public Health. 2025 Mar 6;13:1537742. doi: 10.3389/fpubh.2025.1537742. eCollection 2025. Front Public Health. 2025. PMID: 40115344 Free PMC article.
-
An Experimental Analysis of Five Household Equipment-Based Methods for Decontamination and Reuse of Surgical Masks.Int J Environ Res Public Health. 2022 Mar 11;19(6):3296. doi: 10.3390/ijerph19063296. Int J Environ Res Public Health. 2022. PMID: 35328984 Free PMC article.
References
-
- National Institute for Occupational Safety and Health (NIOSH) Cfr 84 Respiratory Protective Devices: Final Rules and Notice. Federal Register. Federal Register US Centers for Disease Control and Prevention, National Institute for Occu-Pational Safety and Health; Atlanta, GA, USA: 1997.
-
- Centers for Disease Control and Prevention (CDC) Niosh-Approved N95 Particulate Filtering Facepiece Respirators. [(accessed on 21 January 2021)]; Available online: https://www.cdc.gov/niosh/npptl/topics/respirators/disp_part/n95list1.Html.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

