Dynamic flow priming programs allow tuning up the cell layers properties for engineered vascular graft
- PMID: 34282200
- PMCID: PMC8290030
- DOI: 10.1038/s41598-021-94023-9
Dynamic flow priming programs allow tuning up the cell layers properties for engineered vascular graft
Abstract
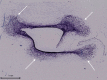
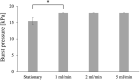
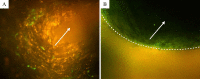
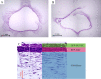
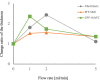
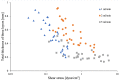
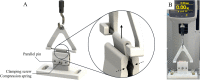
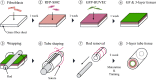
Tissue engineered vascular grafts (TEVG) are potentially clear from ethical and epidemiological concerns sources for reconstructive surgery for small diameter blood vessels replacement. Here, we proposed a novel method to create three-layered TEVG on biocompatible glass fiber scaffolds starting from flat sheet state into tubular shape and to train the resulting tissue by our developed bioreactor system. Constructed tubular tissues were matured and trained under 3 types of individual flow programs, and their mechanical and biological properties were analyzed. Training in the bioreactor significantly increased the tissue burst pressure resistance (up to 18 kPa) comparing to untrained tissue. Fluorescent imaging and histological examination of trained vascular tissue revealed that each cell layer has its own individual response to training flow rates. Histological analysis suggested reverse relationship between tissue thickness and shear stress, and the thickness variation profiles were individual between all three types of cell layers. Concluding: a three-layered tissue structure similar to physiological can be assembled by seeding different cell types in succession; the following training of the formed tissue with increasing flow in a bioreactor is effective for promoting cell survival, improving pressure resistance, and cell layer formation of desired properties.
© 2021. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures







Similar articles
-
A dynamically cultured collagen/cells-incorporated elastic scaffold for small-diameter vascular grafts.J Biomater Sci Polym Ed. 2012;23(14):1807-20. doi: 10.1163/156856211X598201. Epub 2012 May 8. J Biomater Sci Polym Ed. 2012. PMID: 21943800
-
Rapid Self-Assembly of Bioengineered Cardiovascular Bypass Grafts From Scaffold-Stabilized, Tubular Bilevel Cell Sheets.Circulation. 2018 Nov 6;138(19):2130-2144. doi: 10.1161/CIRCULATIONAHA.118.035231. Circulation. 2018. PMID: 30474423 Free PMC article.
-
Physiologic pulsatile flow bioreactor conditioning of poly(ethylene glycol)-based tissue engineered vascular grafts.Ann Biomed Eng. 2007 Feb;35(2):190-200. doi: 10.1007/s10439-006-9099-3. Epub 2006 Dec 16. Ann Biomed Eng. 2007. PMID: 17180465
-
Cell-seeding techniques in vascular tissue engineering.Tissue Eng Part B Rev. 2010 Jun;16(3):341-50. doi: 10.1089/ten.TEB.2009.0527. Tissue Eng Part B Rev. 2010. PMID: 20085439 Free PMC article. Review.
-
Tissue engineered small-diameter vascular grafts.Clin Plast Surg. 2003 Oct;30(4):507-17. doi: 10.1016/s0094-1298(03)00069-5. Clin Plast Surg. 2003. PMID: 14621299 Review.
References
-
- Baquey C. Developments towards tissue-arterial substitutes. Tissue Eng. 2008;5(3):337–347. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

