Asymmetric Radical Process for General Synthesis of Chiral Heteroaryl Cyclopropanes
- PMID: 34282613
- PMCID: PMC8399893
- DOI: 10.1021/jacs.1c04655
Asymmetric Radical Process for General Synthesis of Chiral Heteroaryl Cyclopropanes
Abstract
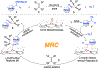
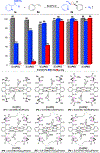
A highly efficient catalytic method has been developed for asymmetric radical cyclopropanation of alkenes with in situ-generated α-heteroaryldiazomethanes via Co(II)-based metalloradical catalysis (MRC). Through fine-tuning the cavity-like environments of newly-synthesized D2-symmetric chiral amidoporphyrins as the supporting ligand, the optimized Co(II)-based metalloradical system is broadly applicable to α-pyridyl and other α-heteroaryldiazomethanes for asymmetric cyclopropanation of wide-ranging alkenes, including several types of challenging substrates. This new catalytic methodology provides a general access to valuable chiral heteroaryl cyclopropanes in high yields with excellent both diastereoselectivities and enantioselectivities. Combined computational and experimental studies further support the underlying stepwise radical mechanism of the Co(II)-based olefin cyclopropanation involving α- and γ-metalloalkyl radicals as the key intermediates.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

Similar articles
-
Metalloradical Catalysis: General Approach for Controlling Reactivity and Selectivity of Homolytic Radical Reactions.Angew Chem Int Ed Engl. 2024 May 13;63(20):e202320243. doi: 10.1002/anie.202320243. Epub 2024 Apr 3. Angew Chem Int Ed Engl. 2024. PMID: 38472114 Free PMC article. Review.
-
Asymmetric Radical Cyclopropanation of Alkenes with In Situ-Generated Donor-Substituted Diazo Reagents via Co(II)-Based Metalloradical Catalysis.J Am Chem Soc. 2017 Jan 25;139(3):1049-1052. doi: 10.1021/jacs.6b11336. Epub 2017 Jan 12. J Am Chem Soc. 2017. PMID: 28051870 Free PMC article.
-
Metalloradical Activation of In Situ-Generated α-Alkynyldiazomethanes for Asymmetric Radical Cyclopropanation of Alkenes.J Am Chem Soc. 2022 Feb 9;144(5):2368-2378. doi: 10.1021/jacs.1c13154. Epub 2022 Jan 31. J Am Chem Soc. 2022. PMID: 35099966 Free PMC article.
-
New Catalytic Radical Process Involving 1,4-Hydrogen Atom Abstraction: Asymmetric Construction of Cyclobutanones.J Am Chem Soc. 2021 Aug 4;143(30):11670-11678. doi: 10.1021/jacs.1c04968. Epub 2021 Jul 22. J Am Chem Soc. 2021. PMID: 34292709 Free PMC article.
-
'Carbene radicals' in Co(II)(por)-catalyzed olefin cyclopropanation.J Am Chem Soc. 2010 Aug 11;132(31):10891-902. doi: 10.1021/ja103768r. J Am Chem Soc. 2010. PMID: 20681723
Cited by
-
A biomimetic SH2 cross-coupling mechanism for quaternary sp3-carbon formation.Science. 2021 Dec 3;374(6572):1258-1263. doi: 10.1126/science.abl4322. Epub 2021 Nov 11. Science. 2021. PMID: 34762491 Free PMC article.
-
Decarboxylative tandem C-N coupling with nitroarenes via SH2 mechanism.Nat Commun. 2022 May 4;13(1):2432. doi: 10.1038/s41467-022-30176-z. Nat Commun. 2022. PMID: 35508545 Free PMC article.
-
Asymmetric Radical Bicyclization for Stereoselective Construction of Tricyclic Chromanones and Chromanes with Fused Cyclopropanes.J Am Chem Soc. 2023 May 31;145(21):11622-11632. doi: 10.1021/jacs.3c01618. Epub 2023 May 2. J Am Chem Soc. 2023. PMID: 37129381 Free PMC article.
-
Transition Metal Catalysis Controlled by Hydrogen Bonding in the Second Coordination Sphere.Chem Rev. 2022 Jul 27;122(14):12308-12369. doi: 10.1021/acs.chemrev.1c00862. Epub 2022 May 20. Chem Rev. 2022. PMID: 35593647 Free PMC article. Review.
-
Metalloradical Catalysis: General Approach for Controlling Reactivity and Selectivity of Homolytic Radical Reactions.Angew Chem Int Ed Engl. 2024 May 13;63(20):e202320243. doi: 10.1002/anie.202320243. Epub 2024 Apr 3. Angew Chem Int Ed Engl. 2024. PMID: 38472114 Free PMC article. Review.
References
-
- For selected books, see:Zard SZ Radical Reactions in Organic Synthesis; Oxford University Press: 2003.
- Chatgilialoglu C; Studer A Encyclopedia of Radicals in Chemistry, Biology, and Materials; John Wiley & Sons: 2012.
- For selected reviews, see:Zard SZ Recent Progress in the Generation and Use of Nitrogen-Centred Radicals. Chem. Soc. Rev 2008, 37, 1603–1618. - PubMed
- Narayanam JM; Stephenson CR Visible Light Photoredox Catalysis: Applications in Organic Synthesis. Chem. Soc. Rev 2011, 40, 102–113. - PubMed
- Quiclet-Sire B; Zard SZ Fun with Radicals: Some New Perspectives for Organic Synthesis. Pure Appl. Chem 2010, 83, 519–551.
- Prier CK; Rankic DA; MacMillan DW Visible Light Photoredox Catalysis with Transition Metal Complexes: Applications in Organic Synthesis. Chem. Rev 2013, 113, 5322–5363. - PMC - PubMed
- Studer A; Curran DP Catalysis of Radical Reactions: A Radical Chemistry Perspective. Angew. Chem., Int. Ed 2016, 55, 58–102. - PubMed
-
- For selected reviews, see:Bar G; Parsons AF Stereoselective Radical Reactions. Chem. Soc. Rev 2003, 32, 251–263. - PubMed
- Sibi MP; Manyem S; Zimmerman J Enantioselective Radical Processes. Chem. Rev 2003, 103, 3263–3295. - PubMed
- Brimioulle R; Lenhart D; Maturi MM; Bach T Enantioselective Catalysis of Photochemical Reactions. Angew. Chem., Int. Ed 2015, 54, 3872–3890. - PubMed
-
- For selected examples on approaches to controlling radical reactivity and stereoselectivity, see:Du JN; Skubi KL; Schultz DM; Yoon TP A Dual-Catalysis Approach to Enantioselective [2 + 2] Photocycloadditions Using Visible Light. Science 2014, 344, 392–396. - PMC - PubMed
- Huo H; Shen X; Wang C; Zhang L; Röse P; Chen L-A; Harms K; Marsch M; Hilt G; Meggers E Asymmetric Photoredox Transition-Metal Catalysis Activated by Visible Light. Nature 2014, 515, 100–103. - PubMed
- Kainz QM; Matier CD; Bartoszewicz A; Zultanski SL; Peters JC; Fu GC Asymmetric Copper-Catalyzed C–N Cross-Couplings Induced by Visible Light. Science 2016, 351, 681–684. - PMC - PubMed
- Zhang W; Wang F; McCann SD; Wang DH; Chen PH; Stahl SS; Liu GS Enantioselective Cyanation of Benzylic C–H Bonds via Copper-Catalyzed Radical Relay. Science 2016, 353, 1014–1018. - PMC - PubMed
- Kern N; Plesniak MP; McDouall JJW; Procter DJ Enantioselective Cyclizations and Cyclization Cascades of Samarium Ketyl Radicals. Nat. Chem 2017, 9, 1198–1204. - PubMed
- Morrill C; Jensen C; Just-Baringo X; Grogan G; Turner NJ; Procter DJ Biocatalytic Conversion of Cyclic Ketones Bearing -Quaternary Stereocenters into Lactones in an Enantioselective Radical Approach to Medium-Sized Carbocycles. Angew. Chem., Int. Ed 2018, 57, 3692–3696. - PMC - PubMed
- Proctor RSJ; Davis HJ; Phipps RJ Catalytic Enantioselective Minisci-Type Addition to Heteroarenes. Science 2018, 360, 419–422. - PubMed
- Biegasiewicz KF; Cooper SJ; Gao X; Oblinsky DG; Kim JH; Garfinkle SE; Joyce LA; Sandoval BA; Scholes GD; Hyster TK Photoexcitation of Favoenzymes Enables a Stereoselective Radical Cyclization. Science 2019, 364, 1166–1169. - PMC - PubMed
- Huang H-M; McDouall JJW; Procter DJ SmI2-Catalysed Cyclization Cascades by Radical Relay. Nat. Catal 2019, 2, 211–218.
- Nakafuku KM; Zhang Z; Wappes EA; Stateman LM; Chen AD; Nagib DA Enantioselective Radical C–H Amination for the Synthesis of β-Amino Alcohols. Nat. Chem 2020, 12, 697–704. - PMC - PubMed
-
- For selected reviews and highlights on Co(II)-based MRC, see:Lu HJ; Zhang XP Catalytic C–H Functionalization by Metalloporphyrins: Recent Developments and Future Directions. Chem. Soc. Rev 2011, 40, 1899–1909. - PubMed
- Pellissier H; Clavier H Enantioselective Cobalt-Catalyzed Transformations. Chem. Rev 2014, 114, 2775–2823. - PubMed
- Demarteau J; Debuigne A; Detrembleur C Organocobalt Complexes as Sources of Carbon-Centered Radicals for Organic and Polymer Chemistries. Chem. Rev 2019, 119, 6906–6955. - PubMed
- Huang H-M; Garduño-Castro MH; Morrill C; Procter DJ Catalytic Cascade Reactions by Radical Relay. Chem. Soc. Rev 2019, 48, 4626–4638. - PubMed
- Singh R; Mukherjee A Metalloporphyrin Catalyzed C–H Amination. ACS Catal. 2019, 9, 3604–3617.
-
- For selected examples of Ti(III)-based radical processes, see:Nugent WA; RajanBabu TV Transition-Metal-Centered Radicals in Organic Synthesis. Titanium(III)-Induced Cyclization of Epoxyolefins. J. Am. Chem. Soc 1988, 110, 8561–8562.
- Rajanbabu TV; Nugent WA Selective Generation of Free-Radicals from Epoxides Using a Transition-Metal Radical. A Powerful New Tool for Organic-Synthesis. J. Am. Chem. Soc 1994, 116, 986–997.
- Gansäuer A; Hildebrandt S; Michelmann A; Dahmen T; von Laufenberg D; Kube C; Fianu GD; Flowers RA II Cationic Titanocene(III) Complexes for Catalysis in Single-Electron Steps. Angew. Chem., Int. Ed 2015, 54, 7003–7006. - PubMed
- Funken N; Mühlhaus F; Gansäuer A General, Highly Selective Synthesis of 1,3-and 1,4-Difunctionalized Building Blocks by Regiodivergent Epoxide Opening. Angew. Chem., Int. Ed 2016, 55, 12030–12034. - PubMed
- Hao W; Wu X; Sun JZ; Siu JC; MacMillan SN; Lin S Radical Redox-Relay Catalysis: Formal [3 + 2] Cycloaddition of N-Acylaziridines and Alkenes. J. Am. Chem. Soc 2017, 139, 12141–12144. - PubMed
- Yao CB; Dahmen T; Gansäuer A; Norton J Anti-Markovnikov Alcohols via Epoxide Hydrogenation through Cooperative Catalysis. Science 2019, 364, 764–767. - PubMed
- Ye KY; McCallum T; Lin S Bimetallic Radical Redox-Relay Catalysis for the Isomerization of Epoxides to Allylic Alcohols. J. Am. Chem. Soc 2019, 141, 9548–9554. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

