Muscle-derived exophers promote reproductive fitness
- PMID: 34288362
- PMCID: PMC8339713
- DOI: 10.15252/embr.202052071
Muscle-derived exophers promote reproductive fitness
Abstract
Organismal functionality and reproduction depend on metabolic rewiring and balanced energy resources. However, the crosstalk between organismal homeostasis and fecundity and the associated paracrine signaling mechanisms are still poorly understood. Using Caenorhabditis elegans, we discovered that large extracellular vesicles (known as exophers) previously found to remove damaged subcellular elements in neurons and cardiomyocytes are released by body wall muscles (BWM) to support embryonic growth. Exopher formation (exopheresis) by BWM is sex-specific and a non-cell autonomous process regulated by developing embryos in the uterus. Embryo-derived factors induce the production of exophers that transport yolk proteins produced in the BWM and ultimately deliver them to newly formed oocytes. Consequently, offspring of mothers with a high number of muscle-derived exophers grew faster. We propose that the primary role of muscular exopheresis is to stimulate reproductive capacity, thereby influencing the adaptation of worm populations to the current environmental conditions.
Keywords: exophers; intertissue signaling; muscle; vesicular transport; vitellogenin.
© 2021 The Authors. Published under the terms of the CC BY 4.0 license.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures

Muscular exophers contain organelles and large protein complexes. Arrows: white—exopher, blue—muscle cell, green—mitochondria, red—proteasome foci. MOM—mitochondrial outer membrane.
BWM actively releases significant amounts of exophers. The image shows the middle part of the worm's body with muscles marked with dashed lines. Arrows indicate representative exophers, and the asterisk indicates the position of the vulva.
The ultrastructure of the muscular exopher and its schematic view. Arrows: red—morphologically changed mitochondria inside the exopher, yellow—normal, elongated mitochondria inside the muscle cell. c—cuticle, m—muscle, e—exopher.
Comparison between muscular exopher and coelomocyte. Arrows: white—exopher, blue—muscle, green—coelomocyte.
Exophers are formed via a pinching‐off mechanism. Arrows: white—exopher, blue—muscle cell, red—distorted muscle cell membrane during exopher formation.
Production of muscular exophers is not evenly distributed across all muscle cells. The highest number of exophers is produced by the muscles adjacent to the vulva. n = 46; N = 3.
Exophers may remain connected to the sending BWM cells via thin elastic tubes that allow further transfer of cellular material. Arrows: white—exopher, blue—muscle cell, red—cellular material transferred to exopher via elastic tube.
Knockdown of two autophagy genes, atg‐7 and lgg‐1 significantly reduces the number of generated exophers. n = 91–103; N = 3.
Proteostasis disruption by hsf‐1 knockdown does not increase exopher production. n = 60 and 55; N = 2.
Challenging proteostasis via oxidative stress but not heat stress increases exophers production. n = 30; N = 3.

- A
Small fraction of muscular exophers contain mitochondria. n = 60; N = 6. n represents the number of worms; N represents the number of experimental repeats that were combined into a single value.
- B
The formation of muscle‐derived exophers, like that of neuronal exophers, is affected by the depletion of EMB‐8 and POD‐1. n = 80–120; pod‐1 RNAi – N = 2, emb‐8 RNAi – N = 3. Data are shown as mean ± SEM; n represents the number of worms; N represents the number of experimental repeats combined into a single value; ****P < 0.0001, Mann–Whitney test.
- C, D
Exophers visualization using correlative light electron microscopy (CLEM). Exopher's red fluorescence (red arrow). (C′, D′) Exopher (red arrow) surrounded by marks (asterisk) after NIRB, autofluorescence of branded frame edges is visible. (C″, D″) CLEM tracking the exopher—LM image, EM image, and their overlay. (C‴, D‴) Image shows exopher's (colored in red) ultrastructure.
The highest number of exophers is produced during the hermaphrodite reproductive period and in aging animals. Males do not produce exophers during the first days of adulthood and begin to generate a small number of exophers later in life. Starting n = 90 hermaphrodites and 150 males; N = 3.
Feminized hermaphrodites of a thermosensitive fem‐1 mutant strain do not produce exophers regardless of growth temperature. This phenotype can be partially rescued by mating fem‐1 mutants with males. n = 10–26; N = 2.
Representative images of the middle part of the worm body in panel B.
Hermaphrodites sterilized via FUdR treatment produce no exophers or only a few per animal. n = 118 and 112 animals; N = 3.
Males and sterile hermaphrodites (via FUdR treatment) show the formation of spherical structures in the BWM that resemble mature exophers. MOM—mitochondrial outer membrane.
The number of produced exophers positively correlates with the number of in utero embryos. The violet line is a linear regression line, and each orange point represents one animal. All animals were 1–3 days old; n = 54; N = 3.
RNAi knockdown of genes regulating the egg‐laying rate and their presence in the uterus influences exopher production. n = 50–60; N = 2.
Embryos retention in the uterus caused by starvation during worm's reproductive period increases exopher production. n = 81–90; N = 3.
Schematic representation of the experimental setup for investigating the influence of embryo‐conditioned buffer on exopher production.
Exposure of young adult worms to embryo‐derived substances increases exopher production. n = 48 and 56; N = 3.
Increased eggshell permeability caused by emb‐27 knockdown elevates exopher production. n = 103 and 89; N = 3.
- A
Exposure of young adult hermaphrodites but not males to lysed embryos increases exopher production. Hermaphrodites: n = 57–74; N = 3. Males: n = 90 and 80; N = 2.
- B
Retention of dead embryos in the hermaphrodite uterus does not increase exopher production. n = 112–178; N = 6 and 3.
- C
Representative images of the mid‐body in panel B. Scale bar is 50 µm.
- D
Disturbance of myosin thick filament assembly in the whole animal (unc‐45 RNAi) but not specifically in BWM (unc‐54 RNAi) leads to decreased exopher production. n = 17–121; N = 2 and 3.
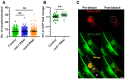
Muscular exophers formation does not depend on the apoptotic engulfment pathway proteins CED‐1 and CED‐6. n = 114–120; N = 3.
CED‐1 depletion leads to a small increase in VIT‐2::GFP in embryos. n = 25 and 26; N = 2.
Images show the formation of an exopher filled with the proteasome and vitellogenin. Images were captured before and after RPN‐5::RFP photobleaching, confirming that the high signal from endogenous VIT‐2::GFP in forming exophers is not an imaging artifact. The white circle marks the bleached area using a 555‐nm laser and the position of the developing exopher. Dashed lines mark different tissue borders: m—muscle, i—intestine, g—gonad. Scale bar is 10 µm.
- A
Schematic representation of the experimental setup for investigating the influence of overactive exopheresis on F0 and F1 worm generation.
- B
Exopher production does not improve muscle functionality. n = 11–38; N = 3.
- C, D
Animals with overactive exopheresis have reduced exploratory behavior presented as a reduction in straight‐line distance traveled (C) and by representative worms traces on the plate (D). n = 11–34; N = 3.
- E
RNAi knockdown of the egg yolk precursor protein VIT‐1 increases the number of muscular exophers. n = 20; N = 2.
- F
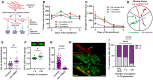
Embryos from hermaphrodite mothers that produce a high number of exophers contain more egg yolk precursor protein VIT‐2. Representative images of embryos with endogenous VIT‐2::GFP levels from mothers with different exopheresis activity are shown above the graph. n = 15 and 14, N = 5 and 6. Scale bar is 10 µm.
- G
RNAi knockdown of RME‐2 yolk receptor abolish exopher production. n = 110–123; N = 3.
- H
Muscle‐produced VIT‐2 is released from muscles via exophers. The image shows the mid‐body of worms expressing the proteasome subunit RPN‐5 tagged with RFP in BWM and VIT‐2::GFP endogenous expression. Arrows: white—exopher, blue—muscle cell. Dashed lines mark embryos present in the uterus. Scale bar is 10 µm.
- I
Offspring of worms with overactive exopheresis develop faster. Y.A.—young adult stage, L4—last larval stage. n = 317–372; N = 2.
Comment in
-
Exophers to feed them all.EMBO Rep. 2021 Aug 4;22(8):e53265. doi: 10.15252/embr.202153265. Epub 2021 Jul 20. EMBO Rep. 2021. PMID: 34288349 Free PMC article.
References
-
- Barral JM, Epstein HF (1999) Protein machines and self assembly in muscle organization. Bioessays 21: 813–823 - PubMed
-
- Bishop D, Nikić I, Brinkoetter M, Knecht S, Potz S, Kerschensteiner M, Misgeld T (2011) Near‐infrared branding efficiently correlates light and electron microscopy. Nat Methods 8: 568–570 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

