Prognostic value of minimal residual disease negativity in myeloma: combined analysis of POLLUX, CASTOR, ALCYONE, and MAIA
- PMID: 34289038
- PMCID: PMC8832474
- DOI: 10.1182/blood.2021011101
Prognostic value of minimal residual disease negativity in myeloma: combined analysis of POLLUX, CASTOR, ALCYONE, and MAIA
Abstract
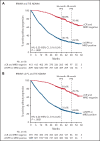
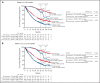
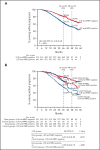
We explored minimal residual disease (MRD) in relapsed/refractory multiple myeloma (RRMM) and transplant-ineligible (TIE) newly diagnosed multiple myeloma (NDMM) using data from 4 phase 3 studies (POLLUX, CASTOR, ALCYONE, and MAIA). Each study previously demonstrated that daratumumab-based therapies improved MRD negativity rates and reduced the risk of disease progression or death by approximately half vs standards of care. We conducted a large-scale pooled analysis for associations between patients achieving complete response or better (≥CR) with MRD-negative status and progression-free survival (PFS). MRD was assessed via next-generation sequencing (10-5 sensitivity threshold). Patient-level data were pooled from all 4 studies and for patients with TIE NDMM and patients with RRMM who received ≤2 prior lines of therapy (≤2 PL). PFS was evaluated by response and MRD status. Median follow-up (months) was 54.8 for POLLUX, 50.2 for CASTOR, 40.1 for ALCYONE, and 36.4 for MAIA. Patients who achieved ≥CR and MRD negativity had improved PFS vs those who failed to reach CR or were MRD positive (TIE NDMM and RRMM hazard ratio [HR] 0.20, P < .0001; TIE NDMM and RRMM ≤2 PL HR 0.20, P < .0001). This benefit occurred irrespective of therapy or disease setting. A time-varying Cox proportional hazard model confirmed that ≥CR with MRD negativity was associated with improved PFS. Daratumumab-based treatment was associated with more patients reaching ≥CR and MRD negativity. These findings represent the first large-scale analysis with robust methodology to support ≥CR with MRD negativity as a prognostic factor for PFS in RRMM and TIE NDMM. These trials were registered at www.clinicaltrials.gov as #NCT02076009, #NCT02136134, #NCT02195479, and #NCT02252172.
© 2022 by The American Society of Hematology.
Figures

Comment in
-
MRD end point in myeloma: ready for prime time?Blood. 2022 Feb 10;139(6):799-802. doi: 10.1182/blood.2021013363. Blood. 2022. PMID: 35142848 No abstract available.
References
-
- Rawstron AC, Child JA, de Tute RM, et al. . Minimal residual disease assessed by multiparameter flow cytometry in multiple myeloma: impact on outcome in the Medical Research Council Myeloma IX Study. J Clin Oncol. 2013;31(20):2540-2547. - PubMed
-
- Lahuerta JJ, Paiva B, Vidriales MB, et al. ; GEM (Grupo Español de Mieloma)/PETHEMA (Programa para el Estudio de la Terapéutica en Hemopatías Malignas) Cooperative Study Group . Depth of response in multiple myeloma: a pooled analysis of three PETHEMA/GEM clinical trials. J Clin Oncol. 2017;35(25):2900-2910. - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

