Cellular dynamics of EMT: lessons from live in vivo imaging of embryonic development
- PMID: 34294089
- PMCID: PMC8296657
- DOI: 10.1186/s12964-021-00761-8
Cellular dynamics of EMT: lessons from live in vivo imaging of embryonic development
Abstract
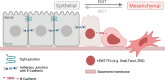
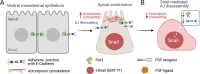
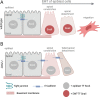
Epithelial-mesenchymal transition (EMT) refers to a process in which epithelial cells lose apical-basal polarity and loosen cell-cell junctions to take on mesenchymal cell morphologies and invasive properties that facilitate migration through extracellular matrix. EMT-and the reverse mesenchymal-epithelial transition (MET)-are evolutionarily conserved processes that are used throughout embryonic development to drive tissue morphogenesis. During adult life, EMT is activated to close wounds after injury, but also can be used by cancers to promote metastasis. EMT is controlled by several mechanisms that depend on context. In response to cell-cell signaling and/or interactions with the local environment, cells undergoing EMT make rapid changes in kinase and adaptor proteins, adhesion and extracellular matrix molecules, and gene expression. Many of these changes modulate localization, activity, or expression of cytoskeletal proteins that mediate cell shape changes and cell motility. Since cellular changes during EMT are highly dynamic and context-dependent, it is ideal to analyze this process in situ in living organisms. Embryonic development of model organisms is amenable to live time-lapse microscopy, which provides an opportunity to watch EMT as it happens. Here, with a focus on functions of the actin cytoskeleton, I review recent examples of how live in vivo imaging of embryonic development has led to new insights into mechanisms of EMT. At the same time, I highlight specific developmental processes in model embryos-gastrulation in fly and mouse embryos, and neural crest cell development in zebrafish and frog embryos-that provide in vivo platforms for visualizing cellular dynamics during EMT. In addition, I introduce Kupffer's vesicle in the zebrafish embryo as a new model system to investigate EMT and MET. I discuss how these systems have provided insights into the dynamics of adherens junction remodeling, planar cell polarity signaling, cadherin functions, and cytoskeletal organization during EMT, which are not only important for understanding development, but also cancer progression. These findings shed light on mechanisms of actin cytoskeletal dynamics during EMT, and feature live in vivo imaging strategies that can be exploited in future work to identify new mechanisms of EMT and MET. Video Abstract.
Keywords: Actin cytoskeleton; Cancer metastasis; Cell migration; Embryonic development; Epithelial-mesenchymal transition (EMT); Gastrulation; In vivo live imaging; Kupffer’s vesicle; Mesenchymal-epithelial transition (MET); Neural crest cell development.
© 2021. The Author(s).
Conflict of interest statement
The author declares he has no competing interests.
Figures



References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous

