Extracellular vesicles in the development of organ-specific metastasis
- PMID: 34295457
- PMCID: PMC8287318
- DOI: 10.1002/jev2.12125
Extracellular vesicles in the development of organ-specific metastasis
Abstract
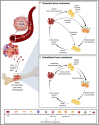
Distant organ metastasis, often termed as organotropic metastasis or metastatic organotropism, is a fundamental feature of malignant tumours and accounts for most cancer-related mortalities. This process is orchestrated by many complex biological interactions and processes that are mediated by a combination of anatomical, genetic, pathophysiological and biochemical factors. Recently, extracellular vesicles (EVs) are increasingly being demonstrated as critical mediators of bi-directional tumour-host cell interactions, controlling organ-specific infiltration, adaptation and colonization at the secondary site. EVs govern organotropic metastasis by modulating the pre-metastatic microenvironment through upregulation of pro-inflammatory gene expression and immunosuppressive cytokine secretion, induction of phenotype-specific differentiation and recruitment of specific stromal cell types. This review discusses EV-mediated metastatic organotropism in visceral (brain, lung, liver, and lymph node) and skeletal (bone) metastasis, and discusses how the pre-metastatic education by EVs transforms the organ into a hospitable, tumour cell-friendly milieu that supports the growth of metastatic cells. Decoding the organ-specific traits of EVs and their functions in organotropic metastasis is essential in accelerating the clinical application of EVs in cancer management.
Keywords: Cancer; extracellular vesicles; intercellular communication; metastasis; organotropism.
© 2021 The Authors. Journal of Extracellular Vesicles published by Wiley Periodicals, LLC on behalf of the International Society for Extracellular Vesicles.
Conflict of interest statement
The authors have declared no conflict of interest.
Figures


References
-
- Abbott, N. J. (2013). Blood‐brain barrier structure and function and the challenges for CNS drug delivery. Journal of Inherited Metabolic Disease, 36(3), 437–449. - PubMed
-
- Ashiru, O. , Boutet, P. , Fernández‐Messina, L. , Agüera‐González, S. , Skepper, J. N. , Valés‐Gómez, M. , & Reyburn, H. T. (2010). Natural killer cell cytotoxicity is suppressed by exposure to the human NKG2D ligand MICA*008 that is shed by tumor cells in exosomes. Cancer Research, 70(2), 481–489. - PMC - PubMed
-
- Atkin‐Smith, G. K. , & Poon, I. K.H. (2017). Disassembly of the dying: Mechanisms and functions. Trends in Cell Biology, 27(2), 151–162. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

