Microglia activation in postmortem brains with schizophrenia demonstrates distinct morphological changes between brain regions
- PMID: 34297453
- PMCID: PMC8713533
- DOI: 10.1111/bpa.13003
Microglia activation in postmortem brains with schizophrenia demonstrates distinct morphological changes between brain regions
Abstract
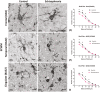
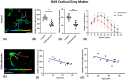
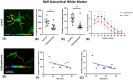
Schizophrenia (SCZ) is a psychiatric disorder that can include symptoms of disorganized speech and thoughts with uncertain underlying mechanisms possibly linked to over-activated microglia. In this study, we used brain samples from sixteen donors with SCZ and thirteen control donors to assess the differential activation of microglia by quantifying density and 3D reconstruction of microglia stained with ionized calcium-binding adaptor molecule-1 (Iba1). Our samples consisted of sections from the frontal, temporal, and cingulate cortical gray matter, subcortical white matter regions (SCWM), and included the anterior corpus callosum. In the first series of studies, we performed a density analysis followed by a spatial analysis to ascertain the microglial density, distribution, and soma size in SCZ brains. Second, we performed a series of morphological quantification techniques to investigate the arborization patterns of the microglia in SCZ. The results demonstrated an increase in microglia density in the cortical gray matter regions in SCZ cases, while in the SCWM, there was a significant increase in microglia density in the frontal and temporal, but not in the other brain regions of interest (ROIs). Spatial analysis using the "nearest neighbor" demonstrated that there was no effect in "clustering", but there were shorter distances between microglia seen in the SCZ cases. The morphological measures showed that there was a region-dependent increase in the microglia soma size in the SCZ cases while the Sholl analysis revealed a significant decrease in the microglia arborization in the SCZ cases across all the ROI's studied. An in-depth 3D reconstruction of microglia in Brodmann area 9 cortical region found that there was a significant association between age and reduced microglial arborization in the SCZ cases. This region-dependent age association can help determine whether longitudinal changes in microglial activation across age are brain region-dependent, which may point to potential therapeutic targets.
Keywords: microglia; morphology; neuroinflammation; postmortem; schizophrenia; spatial analysis.
© 2021 The Authors. Brain Pathology published by John Wiley & Sons Ltd on behalf of International Society of Neuropathology.
Conflict of interest statement
The authors have no duality or conflicts of interest to declare.
Figures




References
-
- Tandon R, Gaebel W, Barch DM, Bustillo J, Gur RE, Heckers S, et al. Definition and description of schizophrenia in the DSM‐5. Schizophr Res. 2013;150(1):3–10. - PubMed
-
- Radewicz K, Garey LJ, Gentleman SM, Reynolds R. Increase in HLA‐DR immunoreactive microglia in frontal and temporal cortex of chronic schizophrenics. J Neuropathol Exp Neurol. 2000;59(2):137–50. - PubMed
-
- Bayer TA, Buslei R, Havas L, Falkai P. Evidence for activation of microglia in patients with psychiatric illnesses. Neurosci Lett. 1999;271(2):126–8. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

