Humoral immunity to SARS-CoV-2 and seasonal coronaviruses in children and adults in north-eastern France
- PMID: 34304047
- PMCID: PMC8299153
- DOI: 10.1016/j.ebiom.2021.103495
Humoral immunity to SARS-CoV-2 and seasonal coronaviruses in children and adults in north-eastern France
Abstract
Background: Children are underrepresented in the COVID-19 pandemic and often experience milder disease than adolescents and adults. Reduced severity is possibly due to recent and more frequent seasonal human coronaviruses (HCoV) infections. We assessed the seroprevalence of SARS-CoV-2 and seasonal HCoV specific antibodies in a large cohort in north-eastern France.
Methods: In this cross-sectional seroprevalence study, serum samples were collected from children and adults requiring hospital admission for non-COVID-19 between February and August 2020. Antibody responses to SARS-CoV-2 and seasonal HCoV (229E, HKU1, NL63, OC43) were assessed using a bead-based multiplex assay, Luciferase-Linked ImmunoSorbent Assay, and a pseudotype neutralisation assay.
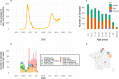
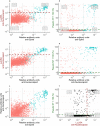
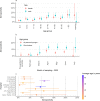
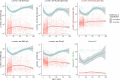
Findings: In 2,408 individuals, seroprevalence of SARS-CoV-2-specific antibodies was 7-8% with three different immunoassays. Antibody levels to seasonal HCoV increased substantially up to the age of 10. Antibody responses in SARS-CoV-2 seropositive individuals were lowest in adults 18-30 years. In SARS-CoV-2 seronegative individuals, we observed cross-reactivity between antibodies to the four HCoV and SARS-CoV-2 Spike. In contrast to other antibodies to SARS-CoV-2, specific antibodies to sub-unit 2 of Spike (S2) in seronegative samples were highest in children. Upon infection with SARS-CoV-2, antibody levels to Spike of betacoronavirus OC43 increased across the whole age spectrum. No SARS-CoV-2 seropositive individuals with low levels of antibodies to seasonal HCoV were observed.
Interpretation: Our findings underline significant cross-reactivity between antibodies to SARS-CoV-2 and seasonal HCoV, but provide no significant evidence for cross-protective immunity to SARS-CoV-2 infection due to a recent seasonal HCoV infection. In particular, across all age groups we did not observe SARS-CoV-2 infected individuals with low levels of antibodies to seasonal HCoV.
Funding: This work was supported by the « URGENCE COVID-19 » fundraising campaign of Institut Pasteur, by the French Government's Investissement d'Avenir program, Laboratoire d'Excellence Integrative Biology of Emerging Infectious Diseases (Grant No. ANR-10-LABX-62-IBEID), and by the REACTing (Research & Action Emerging Infectious Diseases), and by the RECOVER project funded by the European Union's Horizon 2020 research and innovation programme under grant agreement No. 101003589, and by a grant from LabEx IBEID (ANR-10-LABX-62-IBEID).
Keywords: COVID-19; SARS-CoV-2; antibody response; seasonal coronaviruses; sero-epidemiology; seroprevalence.
Copyright © 2021 The Author(s). Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest MTW and SPel are inventors on provisional patent PCT/US 63/057.471 on a serological antibody-based diagnostics of SARS-CoV-2 infection. Dr. Dubos reports grants from Universite de Lille, during the conduct of the study. Dr. van der WERF reports grants from Agence Nationale de la Recherche, grants from European Union's Horizon 2020 research and innovation programme, during the conduct of the study; In addition, Dr. van der WERF has a patent USE OF PROTEINS AND PEPTIDES CODED BY THE GENOME OF A NOVEL STRAIN OF SARS ASSOCIATED CORONAVIRUS issued, and a patent SEVERE ACUTE RESPIRATORY SYNDROME (SARS) - ASSOCIATED CORONAVIRUS DIAGNOSTICS pending.
Figures

References
-
- Organization WH. Coronavirus disease 2019 (COVID-19): situation report. 2021.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

