Utilization of electronic patient-reported outcome measures in cystic fibrosis research: Application to the GALAXY study
- PMID: 34305007
- PMCID: PMC8403637
- DOI: 10.1016/j.jcf.2021.07.002
Utilization of electronic patient-reported outcome measures in cystic fibrosis research: Application to the GALAXY study
Abstract
Background: The Food and Drug Administration considers patient-reported outcome measures (PROMs) an essential part of clinical research studies for approval of new drugs and new indications for existing drugs. GALAXY evaluated the feasibility of electronic PROMs (ePROMS) to conduct a comprehensive evaluation of gastrointestinal (GI) symptoms in persons with cystic fibrosis (pwCF).
Methods: Three validated GI ePROMs (PAC-SYM, PAGI-SYM and PAC-QOL) were combined with a Stool-Specific questionnaire to make up the GALAXY ePROMs and administered prospectively across 26 CF centers in the United States. The ePROMs were completed at enrollment visit and then electronically at weeks 1, 2 and 4. PwCF at least 2 years and older were eligible for the study. Reminders were only provided by the mobile application during the study window.
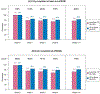
Results: There were 402 participants enrolled in GALAXY. Of those, 169 (42%) were under 18 years old and 193 (48%) were female. The proportion of all follow-up weeks with at least 1 ePROM fully completed was 80%, slightly higher in those ≥18 years of age (82.5%) compared to those <18 years of age (76.5%). When assessing the completion for all 4 ePROMs, the percentage was 77.6%, also higher among those ≥18 year of age (81.5% versus 72.2% for < 18 years of age).
Conclusion: Using ePROMs, our study demonstrated that GI symptoms can be feasibly collected with good reproducibility and with minimal involvement of research coordinator time. This mechanism of symptom collection may provide an efficient tool for future CF trials.
Keywords: Electronic PROMs; Patient reported outcomes measures; cystic fibrosis; gastrointestinal manifestations of cystic fibrosis.
Copyright © 2021 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Conflict of Interest None of the authors have conflicts of interest related to the material in this paper. All of the authors receive funding from CFF with 5 of the authors being employed by CFF (PTV, UK, SLH, CHG).
Figures


References
-
- Burke LB, Kennedy DL, Miskala PH, Papdopoulos EJ, Trentacosti A The use of patient-reported outcomes measures in the evaluation of medical products for regulatory approval. Clin Pharmacol Ther 2008;84(2):281–283. - PubMed
-
- Wagner EH, Austin BT VKM. Organizing care for patients with chronic illness. Milbank Q 1996;74:511–544. - PubMed
-
- Wagner EH, Austin BT, Davis C, Hindmarsh M, Schaefer J, Bonomi A Improving chronic illness care: Translating evidence into action. Heal Aff 2001;20:64–78. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

