Melatonin Protects Against Mdivi-1-Induced Abnormal Spindle Assembly and Mitochondrial Superoxide Production During Porcine Oocyte Maturation
- PMID: 34307369
- PMCID: PMC8297652
- DOI: 10.3389/fcell.2021.693969
Melatonin Protects Against Mdivi-1-Induced Abnormal Spindle Assembly and Mitochondrial Superoxide Production During Porcine Oocyte Maturation
Abstract
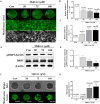
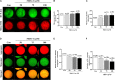
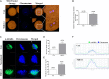
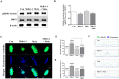
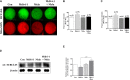
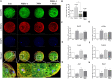
Mitochondrial division inhibitor 1 (Mdivi-1) reportedly provides a close connection between oocyte maturation and mitochondrial function in pigs. N-acetyl-5-methoxy-tryptamine (melatonin) is known to be a representative antioxidant with the ability to rehabilitate meiotic maturation of porcine oocytes. However, the ability of melatonin to recover Mdivi-1-mediated disruption of spindle formation during meiotic maturation of porcine oocytes during in vitro maturation (IVM) has not been studied. Here, we first investigated changes in mitochondrial length, such as fragmentation and elongation form, in mature porcine oocytes during IVM. Mature oocytes require appropriate mitochondrial fission for porcine oocyte maturation. We identified a dose-dependent reduction in meiotic maturation in porcine oocytes following Mdivi-1 treatment (50, 75, and 100 μM). We also confirmed changes in mitochondrial fission protein levels [dynamin-related protein 1 phosphorylation at serine 616 (pDRP1-Ser616) and dynamin-related protein 1 (DRP1)], mitochondrial membrane potential, and ATP production in 75 μM Mdivi-1-treated oocytes. As expected, Mdivi-1 significantly reduced mitochondrial function and DRP1 protein levels and increased spindle abnormalities in porcine oocytes. In addition, we confirmed that melatonin restores abnormal spindle assembly and reduces meiotic maturation rates by Mdivi-1 during porcine oocyte maturation. Interestingly, the expression levels of genes that reduce DNA damage and improve tubulin formation were enhanced during porcine meiotic maturation. Taken together, these results suggest that melatonin has direct beneficial effects on meiotic maturation through tubulin formation factors during porcine oocyte maturation.
Keywords: Mdivi-1; melatonin; mitochondrial fission; pig oocyte maturation; spindle assembly.
Copyright © 2021 Yang, Joe, Bae, Heo, Park and Koo.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



Similar articles
-
Melatonin Improves Oocyte Maturation and Mitochondrial Functions by Reducing Bisphenol A-Derived Superoxide in Porcine Oocytes In Vitro.Int J Mol Sci. 2018 Oct 31;19(11):3422. doi: 10.3390/ijms19113422. Int J Mol Sci. 2018. PMID: 30384504 Free PMC article.
-
The protective role of melatonin in porcine oocyte meiotic failure caused by the exposure to benzo(a)pyrene.Hum Reprod. 2018 Jan 1;33(1):116-127. doi: 10.1093/humrep/dex331. Hum Reprod. 2018. PMID: 29112712
-
Melatonin Promotes in vitro Development of Vitrified-Warmed Mouse GV Oocytes, Potentially by Modulating Phosphorylation of Drp1.Front Vet Sci. 2021 Sep 24;8:752001. doi: 10.3389/fvets.2021.752001. eCollection 2021. Front Vet Sci. 2021. PMID: 34631868 Free PMC article.
-
Melatonin protects against defects induced by malathion during porcine oocyte maturation.J Cell Physiol. 2020 Mar;235(3):2836-2846. doi: 10.1002/jcp.29189. Epub 2019 Sep 18. J Cell Physiol. 2020. PMID: 31535366
-
To mdivi-1 or not to mdivi-1: Is that the question?Dev Neurobiol. 2017 Nov;77(11):1260-1268. doi: 10.1002/dneu.22519. Epub 2017 Aug 30. Dev Neurobiol. 2017. PMID: 28842943 Free PMC article. Review.
Cited by
-
Melatonin as Modulator for Sulfur and Nitrogen Mustard-Induced Inflammation, Oxidative Stress and DNA Damage: Molecular Therapeutics.Antioxidants (Basel). 2023 Feb 6;12(2):397. doi: 10.3390/antiox12020397. Antioxidants (Basel). 2023. PMID: 36829956 Free PMC article. Review.
-
Stabilization of F-Actin Cytoskeleton by Paclitaxel Improves the Blastocyst Developmental Competence through P38 MAPK Activity in Porcine Embryos.Biomedicines. 2022 Aug 2;10(8):1867. doi: 10.3390/biomedicines10081867. Biomedicines. 2022. PMID: 36009414 Free PMC article.
-
Melatonin protects against defects induced by methoxychlor in porcine oocyte maturation.Cell Cycle. 2024 Apr;23(7-8):779-795. doi: 10.1080/15384101.2024.2402190. Epub 2024 Sep 16. Cell Cycle. 2024. PMID: 39285640
-
Nicotinamide mononucleotide biosynthesis and the F-actin cytoskeleton regulate spindle assembly and oocyte maturation quality in post-ovulatory aged porcine oocytes.Cell Commun Signal. 2025 Apr 17;23(1):186. doi: 10.1186/s12964-025-02200-4. Cell Commun Signal. 2025. PMID: 40247324 Free PMC article.
-
Three Classes of Antioxidant Defense Systems and the Development of Postmenopausal Osteoporosis.Front Physiol. 2022 Mar 3;13:840293. doi: 10.3389/fphys.2022.840293. eCollection 2022. Front Physiol. 2022. PMID: 35309045 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

