Cytologic diagnosis of metastatic melanoma by FNA: A practical review
- PMID: 34310059
- PMCID: PMC9292535
- DOI: 10.1002/cncy.22488
Cytologic diagnosis of metastatic melanoma by FNA: A practical review
Abstract
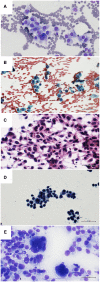
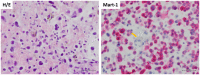
Malignant melanoma (MM) is a highly aggressive neoplasm with a growing worldwide incidence. It is not uncommon that the disease is already metastatic at the time of the first diagnosis. Regional lymph nodes and skin are the first and most common metastatic sites, followed by distant visceral sites (lungs, liver, and central nervous system) and bone. In this clinical setting, fine-needle aspiration (FNA) often represents the first diagnostic approach. FNA is a useful tool to obtain a rapid and accurate diagnosis, in conjunction with ancillary techniques and molecular analysis, as recommended by recent guidelines. The aim of this review was to describe the cytomorphology, immunocytochemical tools, and molecular tools used for the diagnosis of MM metastases on FNA.
Keywords: BRAF; fine-needle aspiration; immunocytochemistry; melanoma; sentinel node.
© 2021 The Authors. Cancer Cytopathology published by Wiley Periodicals LLC on behalf of American Cancer Society.
Conflict of interest statement
The authors made no disclosures.
Figures




Similar articles
-
Cutaneous balloon-cell melanoma metastases to the axillary lymph node: Exploring cytomorphologic features and differential diagnoses on fine needle aspiration biopsy.Cytopathology. 2024 May;35(3):427-431. doi: 10.1111/cyt.13354. Epub 2024 Jan 9. Cytopathology. 2024. PMID: 38196078 No abstract available.
-
Rapid detection of metastatic melanoma in lymph nodes using proton magnetic resonance spectroscopy of fine needle aspiration biopsy specimens.Melanoma Res. 2003 Jun;13(3):259-61. doi: 10.1097/00008390-200306000-00006. Melanoma Res. 2003. PMID: 12777980
-
FNA diagnosis of malignant melanoma-recurrent and metastatic disease.BMJ Case Rep. 2012 Nov 14;2012:bcr2012006887. doi: 10.1136/bcr-2012-006887. BMJ Case Rep. 2012. PMID: 23152178 Free PMC article.
-
Evidence and interdisciplinary consense-based German guidelines: diagnosis and surveillance of melanoma.Melanoma Res. 2007 Dec;17(6):393-9. doi: 10.1097/CMR.0b013e3282f05039. Melanoma Res. 2007. PMID: 17992123
-
[Advancements in the diagnosis of melanoma].Magy Onkol. 2015 Mar;59(1):68-72. Epub 2015 Feb 17. Magy Onkol. 2015. PMID: 25763916 Review. Hungarian.
Cited by
-
PRAME Immunocytochemistry for the Diagnosis of Melanoma Metastases in Cytological Samples.Diagnostics (Basel). 2022 Mar 6;12(3):646. doi: 10.3390/diagnostics12030646. Diagnostics (Basel). 2022. PMID: 35328198 Free PMC article.
-
A novel Cuprotosis-related signature predicts the prognosis and selects personal treatments for melanoma based on bioinformatics analysis.Front Oncol. 2023 Feb 6;13:1108128. doi: 10.3389/fonc.2023.1108128. eCollection 2023. Front Oncol. 2023. PMID: 36824136 Free PMC article.
-
[New aspects in fine needle biopsies of the lymph nodes].Pathologe. 2022 Mar;43(2):109-116. doi: 10.1007/s00292-021-01044-2. Epub 2022 Jan 6. Pathologe. 2022. PMID: 34989818 Review. German.
-
Endobronchial pigmented mass in a patient with primary malignant melanoma of the lung: A case report.Oncol Lett. 2023 Oct 17;26(6):517. doi: 10.3892/ol.2023.14104. eCollection 2023 Dec. Oncol Lett. 2023. PMID: 37927412 Free PMC article.
-
Thermoresponsive M1 macrophage-derived hybrid nanovesicles for improved in vivo tumor targeting.Drug Deliv Transl Res. 2023 Dec;13(12):3154-3168. doi: 10.1007/s13346-023-01378-9. Epub 2023 Jun 26. Drug Deliv Transl Res. 2023. PMID: 37365403 Free PMC article.
References
-
- Cancer Research UK . Cancer Statistics, 2011. Accessed April 30, 2021. http://info.cancerresearchuk.org/cancerstats/
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials

