CAPTURE: a multinational, cross-sectional study of cardiovascular disease prevalence in adults with type 2 diabetes across 13 countries
- PMID: 34315481
- PMCID: PMC8317423
- DOI: 10.1186/s12933-021-01344-0
CAPTURE: a multinational, cross-sectional study of cardiovascular disease prevalence in adults with type 2 diabetes across 13 countries
Abstract
Background: There is a paucity of global data on cardiovascular disease (CVD) prevalence in people with type 2 diabetes (T2D). The primary objective of the CAPTURE study was to estimate the prevalence of established CVD and its management in adults with T2D across 13 countries from five continents. Additional objectives were to further characterize the study sample regarding demographics, clinical parameters and medication usage, with particular reference to blood glucose-lowering agents (GLAs: glucagon-like peptide-1 receptor agonists and sodium-glucose co-transporter-2 inhibitors) with demonstrated cardiovascular benefit in randomized intervention trials.
Methods: Data were collected from adults with T2D managed in primary or specialist care in Australia, China, Japan, Czech Republic, France, Hungary, Italy, Argentina, Brazil, Mexico, Israel, Kingdom of Saudi Arabia, and Turkey in 2019, using standardized methodology. CVD prevalence, weighted by diabetes prevalence in each country, was estimated for the overall CAPTURE sample and participating countries. Country-specific odds ratios for CVD prevalence were further adjusted for relevant demographic and clinical parameters.
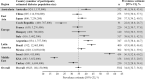
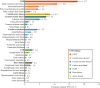
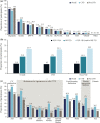
Results: The overall CAPTURE sample included 9823 adults with T2D (n = 4502 from primary care; n = 5321 from specialist care). The overall CAPTURE sample had median (interquartile range) diabetes duration 10.7 years (5.6-17.9 years) and glycated hemoglobin 7.3% (6.6-8.4%) [56 mmol/mol (49-68 mmol/mol)]. Overall weighted CVD and atherosclerotic CVD prevalence estimates were 34.8% (95% confidence interval [CI] 32.7-36.8) and 31.8% (95% CI 29.7-33.8%), respectively. Age, gender, and clinical parameters accounted for some of the between-country variation in CVD prevalence. GLAs with demonstrated cardiovascular benefit were used by 21.9% of participants, which was similar in participants with and without CVD: 21.5% and 22.2%, respectively.
Conclusions: In 2019, approximately one in three adults with T2D in CAPTURE had diagnosed CVD. The low use of GLAs with demonstrated cardiovascular benefit even in participants with established CVD suggested that most were not managed according to contemporary diabetes and cardiology guidelines. Study registration NCT03786406 (registered on December 20, 2018), NCT03811288 (registered on January 18, 2019).
Keywords: Atherosclerotic cardiovascular disease; Cardiovascular disease; Glucagon-like peptide-1 receptor agonists; Non-interventional study; Prevalence; Sodium-glucose co-transporter-2 inhibitors; Type 2 diabetes.
© 2021. The Author(s).
Conflict of interest statement
Ofri Mosenzon reports advisory board membership from Novo Nordisk, Eli Lilly, Sanofi, Merck Sharp & Dohme, Boehringer Ingelheim, Novartis, AstraZeneca, and BOL Pharma; speaker’s bureau honorarium from AstraZeneca, Novo Nordisk, Eli Lilly, Sanofi, Merck Sharp & Dohme, Boehringer Ingelheim, and Janssen; and research grants from Novo Nordisk and AstraZeneca. Abdullah Alguwaihes reports speaker’s bureau honoraria and advisory board membership from AstraZeneca, Boehringer Ingelheim, Eli Lilly, Novo Nordisk, and Sanofi; in addition to research support from AstraZeneca and Novo Nordisk. Jose Luis Arenas Leon reports advisory board membership from Novo Nordisk, AstraZeneca, Merck Sharp & Dohme, and Novartis; speakers bureau honorarium from Novo Nordisk, AstraZeneca, Merck Sharp & Dohme, and Merck; and research grants from Novo Nordisk, AstraZeneca, Amgen, Merck Sharp & Dohme, Novartis, Sanofi, Takeda, Janssen, and Bristol Myers Squibb. Fahri Bayram reports speaker honoraria and travel sponsorship from Bilim İlaç, Abbott, Novo Nordisk, Sanofi Genzyme, Pfizer, and Eczacıbaşı İlaç; advisory board and consultancy for Novo Nordisk, Sanofi Genzyme, Abbott, Pfizer, and Bilim İlaç; and participation in clinical trials for Novo Nordisk, Lilly, Sanofi, and Abbott. Patrice Darmon reports honoraria, consultancy, advisory role, lectures, or travel grants from AstraZeneca, Boehringer Ingelheim, Eli Lilly, Merck Sharp and Dohme, Novartis, Novo Nordisk, Sanofi, Mundipharma, and Abbott. Timothy M.E. Davis reports speaker’s bureau honoraria and advisory board membership from Eli Lilly, Merck Sharp & Dohme, Novo Nordisk, and Sanofi; and speaker’s bureau honorarium from Boehringer Ingelheim and AstraZeneca. Guillermo Dieuzeide reports speaker`s bureau honoraria, advisory board membership, and grants from AstraZeneca, Novo Nordisk, Sanofi Aventis, and Roche Diagnosis. Kristen T. Eriksen Margit S. Kaltoft and Nicolai A. Rhee are employees of Novo Nordisk; Margit S. Kaltoft and Nicolai A. Rhee also own stock and/or shares in Novo Nordisk. Tianpei Hong reports speaker’s bureau honoraria and advisory board membership from AstraZeneca, Boehringer Ingelheim, Eli Lilly, Merck Serono, Merck Sharp & Dohme, Novartis, Novo Nordisk, and Sanofi. Csaba Lengyel reports honoraria and travel grants from AstraZeneca, Berlin Chemie/A Menarini, Boehringer Ingelheim, EGIS, KRKA, Lilly Hungária Kft., MSD Pharma, Novartis, Novo Nordisk, Pfizer, Richter Gedeon, Sandoz, Sanofi Aventis, Servier, TEVA, and Wörwag Pharma. Giuseppina T. Russo reports speaker’s bureau honoraria and advisory board membership from AstraZeneca, Boehringer Ingelheim, Eli Lilly, Merck Sharp & Dohme, Novartis, Novo Nordisk, and Sanofi. Shinichiro Shirabe reports honoraria from Novo Nordisk Pharma Ltd., Sumitomo Dainippon Pharma, and Merck Sharp & Dohme. Katherine Urbancova has worked on oral presentations, articles, and advisory boards for Novo Nordisk, Sanofi Aventis, Boehringer Ingelheim, Novartis, Eli Lilly, and Merck. Sergio Vencio reports research or educational grants from Novartis, Novo Nordisk, Boehringer Ingelheim, and Biomm.
Figures
References
-
- International Diabetes Federation: IDF Diabetes Atlas, 9th edn. Brussels, Belgium: International Diabetes Federation; 2019.
-
- Sarwar N, Gao P, Seshasai SR, Gobin R, Kaptoge S, Di Angelantonio E, et al. Diabetes mellitus, fasting blood glucose concentration, and risk of vascular disease: a collaborative meta-analysis of 102 prospective studies. Lancet. 2010;375:2215–2222. doi: 10.1016/S0140-6736(10)60484-9. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

