Induction of colorectal carcinogenesis in the C57BL/6J and A/J mouse strains with a reduced DSS dose in the AOM/DSS model
- PMID: 34315530
- PMCID: PMC8317392
- DOI: 10.1186/s42826-021-00096-y
Induction of colorectal carcinogenesis in the C57BL/6J and A/J mouse strains with a reduced DSS dose in the AOM/DSS model
Abstract
Background: Colorectal cancer (CRC) is one of the most frequently diagnosed cancers worldwide and thus mouse models of CRC are of significant value to study the pathogenesis. The Azoxymethane/Dextran sulfate sodium (AOM/DSS) model is a widely used, robust initiation-promotion model for chemical induction of colitis-associated CRC in rodents. However, the dosage of chemicals, treatment regimens and outcome measures vary greatly among studies employing this model. Thus, the aim of this study was to examine an AOM/DSS model involving a reduced (1%) dose of DSS for induction of carcinogenesis in A/J and C57BL/6J (B6) mice.
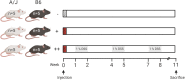
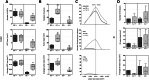
Results: We show that colonic preneoplastic lesions can be reliably detected in A/J and B6 mice by use of a AOM/DSS model involving a single injection of 10 mg/kg AOM followed by three 7-day cycles of a low-dose (1%) DSS administration. Supporting existing evidence of A/J mice exhibiting higher susceptibility to AOM than B6 mice, our AOM/DSS-treated A/J mice developed the highest number of large colonic lesions. Clinical symptoms in both strains subjected to the AOM/DSS treatment did not persist in-between treatment cycles, demonstrating that the animals tolerated the treatment well.
Conclusions: Our findings suggest that a reduced dose of DSS in the AOM/DSS model can be considered in future studies of early phase colorectal carcinogenesis in the A/J and B6 mouse strains using preneoplastic lesions as an outcome measure, and that such regimen may reduce the risk of early trial terminations to accommodate human endpoints. Overall, our data emphasize the importance of devoting attention towards choice of protocol, outcome measures and mouse strain in studies of CRC in mice according to the study purpose.
Keywords: AOM/DSS; Azoxymethane; Colorectal cancer; Dextran sulfate sodium; Disease models; Mouse models.
© 2021. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


References
-
- World Health Organization (WHO): International Agency for Research on Cancer. Globocan 2020: Cancer Fact Sheets — Colorectal Cancer. Available from: https://gco.iarc.fr/today/data/factsheets/cancers/10_8_9-Colorectum-fact....
LinkOut - more resources
Full Text Sources

