Platelet and myeloid cell phenotypes in a rat model of Fabry disease
- PMID: 34320241
- PMCID: PMC8341388
- DOI: 10.1096/fj.202001727RR
Platelet and myeloid cell phenotypes in a rat model of Fabry disease
Abstract
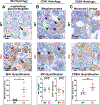
Fabry disease results from a deficiency of the lysosomal enzyme ⍺-Galactosidase-A (⍺-Gal A) and is estimated to occur in approximately 1:4100 live births. Characteristic of the disease is the accumulation of α-Gal-A substrates, primarily the glycosphingolipids (GSLs) globotriaosylceramide and globotriaosylsphingosine. Thrombotic events are a significant concern for Fabry patients, with strokes contributing to a significant decrease in overall lifespan. Currently, the mechanisms underlying the increased risk of thrombotic events experienced by Fabry patients are incompletely defined. Using a rat model of Fabry disease, we provide an improved understanding of the mechanisms linking GSL accumulation to thrombotic risk. We found that ⍺-Gal A-deficient rats accumulate myeloid-derived leukocytes at sites of GSL accumulation, including in the bone marrow and circulation, and that myeloid-derived leukocyte and megakaryocyte populations were prominent among cell types that accumulated GSLs. In the circulation, ⍺-Gal A-deficient rats had increases in cytokine-producing cell types and a corresponding elevation of pro-inflammatory cytokines. Lastly, circulating platelets from ⍺-Gal A-deficient rats accumulated a similar set of ⍺-Galactosidase-A substrates as was observed in megakaryocytes in the bone marrow, and exhibited increased platelet binding to fibrinogen in microfluidic and flow cytometric assays.
Keywords: animal model; glycobiology; glycosphingolipids; lysosomal storage disease.
© 2021 Federation of American Societies for Experimental Biology.
Conflict of interest statement
CONFLICT OF INTEREST
The authors declare no conflicts of interest with the contents of this article.
Figures



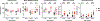



Similar articles
-
Glycosphingolipids and their impact on platelet activity in a murine model of fabry disease.Sci Rep. 2024 Nov 27;14(1):29488. doi: 10.1038/s41598-024-80633-6. Sci Rep. 2024. PMID: 39604471 Free PMC article.
-
α-Galactosidase A-deficient rats accumulate glycosphingolipids and develop cardiorenal phenotypes of Fabry disease.FASEB J. 2019 Jan;33(1):418-429. doi: 10.1096/fj.201800771R. Epub 2018 Jul 6. FASEB J. 2019. PMID: 29979634 Free PMC article.
-
Long-term enzyme correction and lipid reduction in multiple organs of primary and secondary transplanted Fabry mice receiving transduced bone marrow cells.Proc Natl Acad Sci U S A. 2000 Jun 20;97(13):7515-20. doi: 10.1073/pnas.120177997. Proc Natl Acad Sci U S A. 2000. PMID: 10840053 Free PMC article.
-
Progress in the understanding and treatment of Fabry disease.Biochim Biophys Acta Gen Subj. 2020 Jan;1864(1):129437. doi: 10.1016/j.bbagen.2019.129437. Epub 2019 Sep 14. Biochim Biophys Acta Gen Subj. 2020. PMID: 31526868 Free PMC article. Review.
-
Anderson-Fabry disease: a multiorgan disease.Curr Pharm Des. 2013;19(33):5974-96. doi: 10.2174/13816128113199990352. Curr Pharm Des. 2013. PMID: 23448451 Review.
Cited by
-
Platelets in Alcohol-Associated Liver Disease: Interaction With Neutrophils.Cell Mol Gastroenterol Hepatol. 2024;18(1):41-52. doi: 10.1016/j.jcmgh.2024.03.001. Epub 2024 Mar 8. Cell Mol Gastroenterol Hepatol. 2024. PMID: 38461963 Free PMC article. Review.
References
-
- Navarro C, Teijeira S, Dominguez C, Fernandez JM, Rivas E, Fachal C, Barrera S, Rodriguez C, Iranzo P. Fabry disease: an ultrastructural comparative study of skin in hemizygous and heterozygous patients. Acta Neuropathol. 2006;111(2):178–85. Epub 2006/02/08. doi: 10.1007/s00401-005-0026-8. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 DK042667/DK/NIDDK NIH HHS/United States
- R21AI129873/Division of Intramural Research, National Institute of Allergy and Infectious Diseases (DIR, NIAID)
- P41GM103490/HHS | NIH | National Institute of General Medical Sciences (NIGMS)
- K12 HL141954/HL/NHLBI NIH HHS/United States
- R01DK042667/HHS | NIH | National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK)
LinkOut - more resources
Full Text Sources

