An Acceptor-π-Donor Structured Organic Chromophore for NIR Triggered Thermal Ablation of Tumor via DNA Damage-Mediated Apoptosis
- PMID: 34321878
- PMCID: PMC8313434
- DOI: 10.2147/IJN.S319089
An Acceptor-π-Donor Structured Organic Chromophore for NIR Triggered Thermal Ablation of Tumor via DNA Damage-Mediated Apoptosis
Abstract
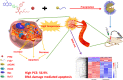
Introduction: It will be challenging to develop high-performance organic chromophores for light-triggered thermal ablation of the tumor. Besides, the mechanisms of organic chromophores for tumor therapy remain unclear. Herein, an acceptor-π-donor (A-π-D) structured organic chromophore based on 2-dicyanomethylenethiazole named PTM was developed for photothermal therapy (PTT) of tumors.
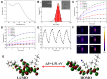
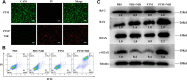
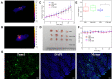
Methods and results: Biocompatible PTM nanoparticles (PTM NPs) were fabricated by enclosing PTM with Pluronic F-127. The results of optical and photothermal properties of PTM NPs showed robust near-infrared (NIR) absorption, excellent photostability and high photothermal conversion efficiency (56.9%). The results of flow cytometry, fluorescence microscopy, apoptosis, CCK-8 assays and animal experiments showed that PTM NPs had a good killing effect on tumors under NIR laser irradiation. Furthermore, mechanistic studies, RNA-seq and biological analysis revealed that PTM NPs can cause tumor cell death via DNA damage-mediated apoptosis.
Conclusion: Light-induced thermal ablation effects of PTM NPs in vitro and vivo were surveyed. Collectively, our studies provided a new approach to developing a safe and effective photothermal agent for cancer treatment.
Keywords: 2-dicyanomethylenethiazole; DNA damage induced apoptosis; NIR absorbing chromophore; RNA-seq; biological analysis; photothermal therapy.
© 2021 Zhang et al.
Conflict of interest statement
The authors report no conflicts of interest in this work.
Figures

Similar articles
-
Dual-Modal Near-Infrared Organic Nanoparticles: Integrating Mild Hyperthermia Phototherapy with Fluorescence Imaging.Int J Nanomedicine. 2024 Sep 5;19:9071-9090. doi: 10.2147/IJN.S472882. eCollection 2024. Int J Nanomedicine. 2024. PMID: 39253059 Free PMC article.
-
Ultrastable Near-Infrared Nonlinear Organic Chromophore Nanoparticles with Intramolecular Charge Transfer for Dually Photoinduced Tumor Ablation.Adv Healthc Mater. 2020 Oct;9(20):e2001042. doi: 10.1002/adhm.202001042. Epub 2020 Sep 16. Adv Healthc Mater. 2020. PMID: 32935929
-
Near-infrared induced phase-shifted ICG/Fe3O4 loaded PLGA nanoparticles for photothermal tumor ablation.Sci Rep. 2017 Jul 14;7(1):5490. doi: 10.1038/s41598-017-06122-1. Sci Rep. 2017. PMID: 28710483 Free PMC article.
-
A thieno-isoindigo derivative-based conjugated polymer nanoparticle for photothermal therapy in the NIR-II bio-window.Nanoscale. 2020 Oct 8;12(38):19665-19672. doi: 10.1039/d0nr03771k. Nanoscale. 2020. PMID: 32966502
-
Aggregation-Enhanced Photophysical Performance of D-π-A Structured Hemicyanine for NIR-II Fluorescent and Photoacoustic Imaging-Guided Photothermal Therapy.Chem Biomed Imaging. 2023 Mar 6;1(6):541-549. doi: 10.1021/cbmi.2c00004. eCollection 2023 Sep 25. Chem Biomed Imaging. 2023. PMID: 39473569 Free PMC article.
Cited by
-
Mitochondria-Targeting Type-I Photodynamic Therapy Based on Phenothiazine for Realizing Enhanced Immunogenic Cancer Cell Death via Mitochondrial Oxidative Stress.Int J Nanomedicine. 2025 Jan 6;20:125-139. doi: 10.2147/IJN.S494970. eCollection 2025. Int J Nanomedicine. 2025. PMID: 39802375 Free PMC article.
-
Chidamide stacked in magnetic polypyrrole nano-composites counter thermotolerance and metastasis for visualized cancer photothermal therapy.Drug Deliv. 2022 Dec;29(1):1312-1325. doi: 10.1080/10717544.2022.2068697. Drug Deliv. 2022. PMID: 35475384 Free PMC article.
References
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

