Mechanical Overloading Induced-Activation of mTOR Signaling in Tendon Stem/Progenitor Cells Contributes to Tendinopathy Development
- PMID: 34322484
- PMCID: PMC8311934
- DOI: 10.3389/fcell.2021.687856
Mechanical Overloading Induced-Activation of mTOR Signaling in Tendon Stem/Progenitor Cells Contributes to Tendinopathy Development
Abstract
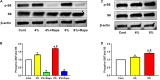
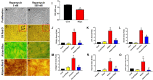
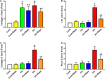
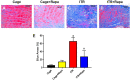
Despite the importance of mechanical loading in tendon homeostasis and pathophysiology, the molecular responses involved in the mechanotransduction in tendon cells remain unclear. In this study, we found that in vitro mechanical loading activated the mammalian target of rapamycin (mTOR) in rat patellar tendon stem/progenitor cells (TSCs) in a stretching magnitude-dependent manner. Application of rapamycin, a specific inhibitor of mTOR, attenuated the phosphorylation of S6 and 4E-BP1 and as such, largely inhibited the mechanical activation of mTOR. Moreover, rapamycin significantly decreased the proliferation and non-tenocyte differentiation of PTSCs as indicated by the reduced expression levels of LPL, PPARγ, SOX-9, collagen II, Runx-2, and osteocalcin genes. In the animal studies, mice subjected to intensive treadmill running (ITR) developed tendon degeneration, as evidenced by the formation of round-shaped cells, accumulation of proteoglycans, and expression of SOX-9 and collagen II proteins. However, daily injections of rapamycin in ITR mice reduced all these tendon degenerative changes. Collectively, these findings suggest that mechanical loading activates the mTOR signaling in TSCs, and rapamycin may be used to prevent tendinopathy development by blocking non-tenocyte differentiation due to mechanical over-activation of mTOR in TSCs.
Keywords: mTOR; mechanical loading; rapamycin; tendon stem cells; treadmill running.
Copyright © 2021 Nie, Zhou, Wang, Zhang and Wang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Moderate and intensive mechanical loading differentially modulate the phenotype of tendon stem/progenitor cells in vivo.PLoS One. 2020 Dec 29;15(12):e0242640. doi: 10.1371/journal.pone.0242640. eCollection 2020. PLoS One. 2020. PMID: 33373386 Free PMC article.
-
The effects of mechanical loading on tendons--an in vivo and in vitro model study.PLoS One. 2013 Aug 19;8(8):e71740. doi: 10.1371/journal.pone.0071740. eCollection 2013. PLoS One. 2013. PMID: 23977130 Free PMC article.
-
Moderate Exercise Mitigates the Detrimental Effects of Aging on Tendon Stem Cells.PLoS One. 2015 Jun 18;10(6):e0130454. doi: 10.1371/journal.pone.0130454. eCollection 2015. PLoS One. 2015. PMID: 26086850 Free PMC article.
-
Production of PGE(2) increases in tendons subjected to repetitive mechanical loading and induces differentiation of tendon stem cells into non-tenocytes.J Orthop Res. 2010 Feb;28(2):198-203. doi: 10.1002/jor.20962. J Orthop Res. 2010. PMID: 19688869
-
Mechanobiology of young and aging tendons: In vivo studies with treadmill running.J Orthop Res. 2018 Feb;36(2):557-565. doi: 10.1002/jor.23761. Epub 2017 Nov 22. J Orthop Res. 2018. PMID: 28976604 Free PMC article. Review.
Cited by
-
Metformin improves tendon degeneration by blocking translocation of HMGB1 and suppressing tendon inflammation and senescence in aging mice.J Orthop Res. 2023 Jun;41(6):1162-1176. doi: 10.1002/jor.25470. Epub 2022 Nov 7. J Orthop Res. 2023. PMID: 36262012 Free PMC article.
-
Comparison of Tendon Development Versus Tendon Healing and Regeneration.Front Cell Dev Biol. 2022 Jan 24;10:821667. doi: 10.3389/fcell.2022.821667. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 35141224 Free PMC article. Review.
-
Erroneous Differentiation of Tendon Stem/Progenitor Cells in the Pathogenesis of Tendinopathy: Current Evidence and Future Perspectives.Stem Cell Rev Rep. 2025 Feb;21(2):423-453. doi: 10.1007/s12015-024-10826-z. Epub 2024 Nov 23. Stem Cell Rev Rep. 2025. PMID: 39579294 Review.
-
Rotator cuff tears.Nat Rev Dis Primers. 2024 Feb 8;10(1):8. doi: 10.1038/s41572-024-00492-3. Nat Rev Dis Primers. 2024. PMID: 38332156 Review.
-
Recent Advances in the Use of Stem Cells in Tissue Engineering and Adjunct Therapies for Tendon Reconstruction and Future Perspectives.Int J Mol Sci. 2024 Apr 19;25(8):4498. doi: 10.3390/ijms25084498. Int J Mol Sci. 2024. PMID: 38674084 Free PMC article. Review.
References
-
- Bastiani G., Corte F. D., Brass K. E., Cantarelli C., Dau S., Kommers G. D., et al. (2018). Histochemistry of equine damaged tendons, ligaments and articular cartilage. Acta Sci. Vet. 146:1612.
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

