Human Platelet Lysate as an Alternative to Autologous Serum for Human Chondrocyte Clinical Use
- PMID: 34330164
- PMCID: PMC8808884
- DOI: 10.1177/19476035211035433
Human Platelet Lysate as an Alternative to Autologous Serum for Human Chondrocyte Clinical Use
Abstract
Objective: A pivotal aspect of cartilage tissue engineering resides in cell culture medium supplementation, in view of maximizing in vitro cell proliferation and preserving cellular functionality. Autologous human serum (aHS) is commonly used as an inducive supplement for safe human articular chondrocyte (HAC) proliferation prior to clinical implantation. However, practical clinical use of aHS is hindered by constraining manufacturing requirements and quality assurance-driven downstream processing. The present study investigated potential alternative use of commercial human platelet lysate (hPL) supplements in HAC manufacturing workflows related to clinical therapeutic pathways.
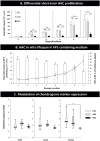
Design: Differential effects of hPL, aHS, and fetal bovine serum were assessed on primary cultured HAC parameters (viability, proliferative rates, and morphology) in 2-dimensional in vitro systems. A 3-dimensional HAC pellet model served for postexpansion assessment of cellular functionality, by visualizing proteoglycan production (Alcian blue staining), and by using qRT-PCR relative quantification of chondrogenic marker (SOX9, COL2-A1, and ACAN) genetic expression.
Results: We found that monolayer HAC culture with hPL or aHS supplements presented similar characteristics (elongated cell morphology and nearly identical growth kinetics). Chondrogenic activity appeared as conserved in HACs expanded with human or bovine supplements, wherein histologic analysis indicated a progressive sGAG accumulation and SOX9, COL2-A1, ACAN gene expression was upregulated in 3-dimensional HAC pellet models.
Conclusion: This study therefore supports the use of hPL as a functional equivalent and alternative to aHS for cultured HAC batch preparation, with the potential to effectively alleviate pressure on clinical and manufacturing bottlenecks in cell therapy approaches for cartilage regeneration.
Keywords: autologous chondrocyte culture; cartilage repair; cell therapy; chondrocyte transplantation; human platelet lysate.
Conflict of interest statement
Figures


References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

