Ginkgo biloba extract protects human neuroblastoma SH-SY5Y cells against oxidative glutamate toxicity by activating redoxosome-p66Shc
- PMID: 34335893
- PMCID: PMC8290427
- DOI: 10.3892/etm.2021.10383
Ginkgo biloba extract protects human neuroblastoma SH-SY5Y cells against oxidative glutamate toxicity by activating redoxosome-p66Shc
Abstract
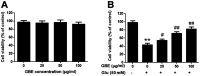
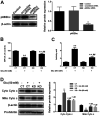
Ginkgo biloba extract (GBE), a traditional Chinese herbal medicine component, is widely used to alleviate symptoms of neurodegenerative diseases. It has been confirmed that GBE exerts its pharmacological effect mainly due to its antioxidant activity; however, the molecular mechanism responsible for this effect remains unclear. The aim of the present study was to investigate the detailed mechanism of GBE, the main component of Gingko biloba dropping medicine, against oxidative glutamate toxicity in human neuroblastoma SH-SY5Y cells. The SH-SY5Y cells were untreated or pretreated with GBE followed by glutamate stimulation. Cell viability was assessed using an MTT assay. In addition, oxidative stress indexes, including intracellular ROS generation and NADPH oxidase and caspase activity, were also measured. The protein expression of key signaling factors involved in the redoxosome-p66Shc pathway was evaluated to elucidate the neuroprotective effect of GBE. The results showed that GBE treatment significantly attenuated the glutamate-induced cytotoxicity in SH-SY5Y cells by suppressing oxidative stress. A mechanical study revealed that redoxosome-p66Shc activation was associated with glutamate-induced cytotoxicity, which caused mitochondrial dysfunction and cell death. Interestingly, GBE treatment attenuated the activation of redoxosome-p66Shc in a dose-dependent manner, which suggested that the protective effect of GBE on SH-SY5Y cells against oxidative glutamate toxicity may be mediated by the modulation of redoxosome-p66Shc signaling. The current findings contribute to a better understanding of the therapeutic effect of GBE and indicate that redoxosome-p66Shc signaling might be a novel therapeutic target in the prevention and/or treatment of neurodegenerative diseases.
Keywords: Ginkgo biloba extract; glutamate; neurodegenerative diseases; redoxosome-p66Shc signaling.
Copyright: © Wang et al.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures







Similar articles
-
Ginkgo biloba Extract Attenuates Light-Induced Photoreceptor Degeneration by Modulating CAV-1-Redoxosome Signaling.Antioxidants (Basel). 2022 Jun 27;11(7):1268. doi: 10.3390/antiox11071268. Antioxidants (Basel). 2022. PMID: 35883759 Free PMC article.
-
Ginkgo biloba extracts protect human retinal Müller glial cells from t-BHP induced oxidative damage by activating the AMPK-Nrf2-NQO-1 axis.J Pharm Pharmacol. 2023 Mar 12;75(3):385-396. doi: 10.1093/jpp/rgac095. J Pharm Pharmacol. 2023. PMID: 36583518
-
Ginkgo biloba extract ameliorates hyperglycaemia-induced enteric glial cell injury via regulation of the TLR2-related pathway.J Pharm Pharmacol. 2023 Nov 23;75(11):1430-1441. doi: 10.1093/jpp/rgad075. J Pharm Pharmacol. 2023. PMID: 37738214
-
Ginkgo biloba Extract in Vascular Protection: Molecular Mechanisms and Clinical Applications.Curr Vasc Pharmacol. 2017;15(6):532-548. doi: 10.2174/1570161115666170713095545. Curr Vasc Pharmacol. 2017. PMID: 28707602 Review.
-
New Insight into the Mechanisms of Ginkgo Biloba Extract in Vascular Aging Prevention.Curr Vasc Pharmacol. 2020;18(4):334-345. doi: 10.2174/1570161117666190621150725. Curr Vasc Pharmacol. 2020. PMID: 31223090 Review.
Cited by
-
Anti-Aging of the Nervous System and Related Neurodegenerative Diseases With Chinese Herbal Medicine.Am J Alzheimers Dis Other Demen. 2023 Jan-Dec;38:15333175231205445. doi: 10.1177/15333175231205445. Am J Alzheimers Dis Other Demen. 2023. PMID: 37818604 Free PMC article. Review.
-
Investigating the effects of Ginkgo biloba leaf extract on cognitive function in Alzheimer's disease.CNS Neurosci Ther. 2024 Sep;30(9):e14914. doi: 10.1111/cns.14914. CNS Neurosci Ther. 2024. PMID: 39238068 Free PMC article.
-
Ginkgo biloba Extract Attenuates Light-Induced Photoreceptor Degeneration by Modulating CAV-1-Redoxosome Signaling.Antioxidants (Basel). 2022 Jun 27;11(7):1268. doi: 10.3390/antiox11071268. Antioxidants (Basel). 2022. PMID: 35883759 Free PMC article.
-
Effects of Different Drying Methods on the Quality of Bletilla striata Scented Tea.Molecules. 2023 Mar 7;28(6):2438. doi: 10.3390/molecules28062438. Molecules. 2023. PMID: 36985409 Free PMC article.
References
-
- Kanazawa I. Neurotransmitters and neurodegenerative disorders. Clin Ther. 1984;7:48–58. - PubMed
LinkOut - more resources
Full Text Sources
