Fusobacterium nucleatum CbpF Mediates Inhibition of T Cell Function Through CEACAM1 Activation
- PMID: 34336716
- PMCID: PMC8319768
- DOI: 10.3389/fcimb.2021.692544
Fusobacterium nucleatum CbpF Mediates Inhibition of T Cell Function Through CEACAM1 Activation
Abstract
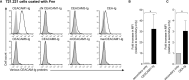
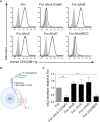
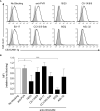
F. nucleatum is an anaerobic bacterium that is associated with several tumor entities and promotes tumorigenesis. Recent evidence suggests that F. nucleatum binds the inhibitory receptor carcinoembryonic antigen cell adhesion molecule 1 (CEACAM1) via the trimeric autotransporter adhesin CbpF. However, whether this binding is functional or whether other fusobacterial trimeric autotransporter adhesins are involved in CEACAM1 activation is unknown. In this study, using F. nucleatum mutants lacking the type 5c trimeric autotransporter adhesins fvcA (CbpF), fvcB, fvcC, and fvcD, we show that F. nucleatum CbpF binds and activates CEACAM1 and also binds carcinoembryonic antigen (CEA), a tumor-associated protein. We further find that CEACAM antibodies directed against the CEACAM N-terminal domain block the CbpF-CEACAM1 interaction. In functional assays, we demonstrate CbpF-dependent inhibition of CD4+ T cell response. Thus, we characterize an immune evasion mechanism in which F. nucleatum uses its surface protein CbpF to inhibit T cell function by activating CEACAM1.
Keywords: CEA; CEACAM1; CbpF; F. nucleatum; trimeric autotransporter adhesins.
Copyright © 2021 Galaski, Shhadeh, Umaña, Yoo, Arpinati, Isaacson, Berhani, Singer, Slade, Bachrach and Mandelboim.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

