ASN007 is a selective ERK1/2 inhibitor with preferential activity against RAS-and RAF-mutant tumors
- PMID: 34337566
- PMCID: PMC8324497
- DOI: 10.1016/j.xcrm.2021.100350
ASN007 is a selective ERK1/2 inhibitor with preferential activity against RAS-and RAF-mutant tumors
Abstract
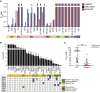
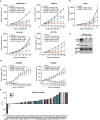
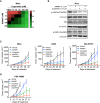
Inhibition of the extracellular signal-regulated kinases ERK1 and ERK2 (ERK1/2) offers a promising therapeutic strategy in cancers harboring activated RAS/RAF/MEK/ERK signaling pathways. Here, we describe an orally bioavailable and selective ERK1/2 inhibitor, ASN007, currently in clinical development for the treatment of cancer. In preclinical studies, ASN007 shows strong antiproliferative activity in tumors harboring mutations in BRAF and RAS (KRAS, NRAS, and HRAS). ASN007 demonstrates activity in a BRAFV600E mutant melanoma tumor model that is resistant to BRAF and MEK inhibitors. The PI3K inhibitor copanlisib enhances the antiproliferative activity of ASN007 both in vitro and in vivo due to dual inhibition of RAS/MAPK and PI3K survival pathways. Our data provide a rationale for evaluating ASN007 in RAS/RAF-driven tumors as well as a mechanistic basis for combining ASN007 with PI3K inhibitors.
Keywords: ASN007; ERK; KRAS; PI3K; RAF/RAS-driven cancers; biomarker; combinational therapy; kinase inhibitor; lymphoma; solid tumors.
© 2021.
Conflict of interest statement
A.Y. has received honoraria and/or consultancy fees from Abbvie, Biopath, Curis, Epizyme, Janssen, Merck, Roche, Takeda, and Xynomic and has received research support from Janssen, Curis, Merck, BMS, Syndax, and Roche. S.T., S.G., and L.D. are employees and shareholders of Asana BioSciences. R.A.S. is a shareholder in and consultant to Asana BioSciences. S.T., R.A.S., and S.R. are inventors on three patents covering ASN007 and related compounds and their use in the treatment of cancer. The other authors declare no competing interests.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

