SPM Receptor Expression and Localization in Irradiated Salivary Glands
- PMID: 34339312
- PMCID: PMC8335844
- DOI: 10.1369/00221554211031678
SPM Receptor Expression and Localization in Irradiated Salivary Glands
Abstract
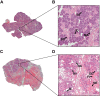
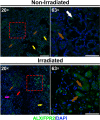
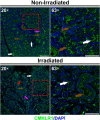
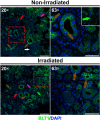
Radiation therapy-mediated salivary gland destruction is characterized by increased inflammatory cell infiltration and fibrosis, both of which ultimately lead to salivary gland hypofunction. However, current treatments (e.g., artificial saliva and sialagogues) only promote temporary relief of symptoms. As such, developing alternative measures against radiation damage is critical for restoring salivary gland structure and function. One promising option for managing radiation therapy-mediated damage in salivary glands is by activation of specialized proresolving lipid mediator receptors due to their demonstrated role in resolution of inflammation and fibrosis in many tissues. Nonetheless, little is known about the presence and function of these receptors in healthy and/or irradiated salivary glands. Therefore, the goal of this study was to detect whether these specialized proresolving lipid mediator receptors are expressed in healthy salivary glands and, if so, if they are maintained after radiation therapy-mediated damage. Our results indicate that specialized proresolving lipid mediator receptors are heterogeneously expressed in inflammatory as well as in acinar and ductal cells within human submandibular glands and that their expression persists after radiation therapy. These findings suggest that epithelial cells as well as resident immune cells represent potential targets for modulation of resolution of inflammation and fibrosis in irradiated salivary glands.
Keywords: fibrosis; inflammation; radiation therapy; specialized proresolving lipid mediators.
Conflict of interest statement
Figures

References
-
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70(1):7–30. - PubMed
-
- Tyler MA, Mohamed ASR, Smith JB, Aymard JM, Fuller CD, Phan J, Frank SJ, Ferrarotto R, Kupferman ME, Hanna EY, Gunn GB, Su SY. Long-term quality of life after definitive treatment of sinonasal and nasopharyngeal malignancies. Laryngoscope. 2020;130(1):86–93. - PubMed
-
- Ge X, Liao Z, Yuan J, Mao D, Li Y, Yu E, Wang X, Ding Z. Radiotherapy-related quality of life in patients with head and neck cancers: a meta-analysis. Support Care Cancer. 2020;28(6):2701–12. - PubMed
-
- Pedersen AM, Bardow A, Jensen SB, Nauntofte B. Saliva and gastrointestinal functions of taste, mastication, swallowing and digestion. Oral Dis. 2002;8(3):117–29. - PubMed
-
- Jehmlich N, Stegmaier P, Golatowski C, Salazar MG, Rischke C, Henke M, Völker U. Differences in the whole saliva baseline proteome profile associated with development of oral mucositis in head and neck cancer patients undergoing radiotherapy. J Proteomics. 2015;125:98–103. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

