Clinical outcomes and outcome measurement tools reported in randomised controlled trials of treatment for snakebite envenoming: A systematic review
- PMID: 34339410
- PMCID: PMC8360524
- DOI: 10.1371/journal.pntd.0009589
Clinical outcomes and outcome measurement tools reported in randomised controlled trials of treatment for snakebite envenoming: A systematic review
Abstract
Background: Snakebite is a priority neglected tropical disease and causes a range of complications that vary depending on the snake species. Randomised clinical trials have used varied outcome measures that do not allow results to be compared or combined. In accordance with the Core Outcomes Measurements in Effectiveness Trials (COMET) initiative, this systematic review aims to support the development of a globally relevant core outcome set for snakebite.
Methods: All randomised controlled trials, secondary analyses of randomised controlled trials and study protocols investigating the efficacy of therapeutics for human snakebite envenoming were eligible for inclusion. Study screening and data extraction were conducted in duplicate by two independent reviewers. All primary and secondary outcome measures were extracted and compiled, as were adverse event outcome measures. Similar outcome measures were grouped into domains. The study was prospectively registered with PROSPERO: CRD42020196160.
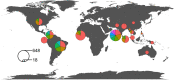
Results: This systematic review included 43 randomised controlled trials, two secondary analyses and 13 study protocols. A total of 382 outcome measures were extracted and, after duplicates were merged, there were 153 unique outcomes. The most frequently used outcome domain ('venom antigenaemia') was included in less than one third of the studies. The unique outcomes were classified into 60 outcome domains. Patient-centred outcomes were used in only three of the studies.
Discussion: Significant heterogeneity in outcome measures exists in snakebite clinical trials. Consensus is needed to select outcome measures that are valid, reliable, patient-centred and feasible. The results of this systematic review strongly support the development of a core outcome set for use in snakebite clinical trials.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



References
-
- Kasturiratne A, Wickremasinghe AR, de Silva N, Gunawardena NK, Pathmeswaran A, Premaratna R, et al.. The Global Burden of Snakebite: A Literature Analysis and Modelling Based on Regional Estimates of Envenoming and Deaths. PLoS Medicine. 2008;5: e218. doi: 10.1371/journal.pmed.0050218 - DOI - PMC - PubMed
-
- Global funding for snakebite envenoming research. [cited 6 Apr 2020]. Available: https://wellcome.ac.uk/sites/default/files/global-funding-for-snakebite-...
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

