Potential protective role of the anti-PD-1 blockade against SARS-CoV-2 infection
- PMID: 34339917
- PMCID: PMC8315943
- DOI: 10.1016/j.biopha.2021.111957
Potential protective role of the anti-PD-1 blockade against SARS-CoV-2 infection
Abstract
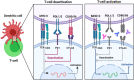
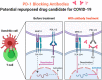
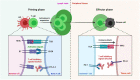
The outbreak of Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in Wuhan, China, in December 2019, and its global dissemination became the coronavirus disease 2019 (COVID-19) pandemic declared by the World Health Organization (WHO) on 11 March 2020. In patients undergoing immunotherapy, the effect and path of viral infection remain uncertain. In addition, viral-infected mice and humans show T-cell exhaustion, which is identified after infection with SARS-CoV-2. Notably, they regain their T-cell competence and effectively prevent viral infection when treated with anti-PD-1 antibodies. Four clinical trials are officially open to evaluate anti-PD-1 antibody administration's effectiveness for cancer and non-cancer individuals influenced by COVID-19 based on these findings. The findings may demonstrate the hypothesis that a winning strategy to combat SARS-CoV-2 infection could be the restoration of exhausted T-cells. In this review, we outline the potential protective function of the anti-PD-1 blockade against SARS-CoV-2 infection with the aim to develop SARS-CoV-2 therapy.
Keywords: Anti-PD-1; COVID-19; Immunotherapy; SARS-CoV-2; T-cells.
Copyright © 2021 The Authors. Published by Elsevier Masson SAS.. All rights reserved.
Conflict of interest statement
The authors have declared that no competing interest exists.
Figures


Similar articles
-
PD-1 blockade counteracts post-COVID-19 immune abnormalities and stimulates the anti-SARS-CoV-2 immune response.JCI Insight. 2021 Dec 22;6(24):e146701. doi: 10.1172/jci.insight.146701. JCI Insight. 2021. PMID: 34784300 Free PMC article.
-
Immune-checkpoint inhibitors from cancer to COVID‑19: A promising avenue for the treatment of patients with COVID‑19 (Review).Int J Oncol. 2021 Feb;58(2):145-157. doi: 10.3892/ijo.2020.5159. Epub 2020 Dec 14. Int J Oncol. 2021. PMID: 33491759 Free PMC article. Review.
-
T-cell responses and therapies against SARS-CoV-2 infection.Immunology. 2021 Jan;162(1):30-43. doi: 10.1111/imm.13262. Epub 2020 Oct 27. Immunology. 2021. PMID: 32935333 Free PMC article. Review.
-
Designing therapeutic strategies to combat severe acute respiratory syndrome coronavirus-2 disease: COVID-19.Drug Dev Res. 2021 Feb;82(1):12-26. doi: 10.1002/ddr.21720. Epub 2020 Jul 30. Drug Dev Res. 2021. PMID: 33216381 Review.
-
Immune checkpoint inhibitors and SARS-CoV2 infection.Int Immunopharmacol. 2024 Aug 20;137:112419. doi: 10.1016/j.intimp.2024.112419. Epub 2024 Jun 11. Int Immunopharmacol. 2024. PMID: 38865755 Review.
Cited by
-
Immune checkpoint inhibitor therapy and outcomes from SARS-CoV-2 infection in patients with cancer: a joint analysis of OnCovid and ESMO-CoCARE registries.J Immunother Cancer. 2022 Nov;10(11):e005732. doi: 10.1136/jitc-2022-005732. J Immunother Cancer. 2022. PMID: 36450384 Free PMC article.
-
Clinical, radiological and pathological findings in patients with persistent lung disease following SARS-CoV-2 infection.Eur Respir J. 2022 Oct 6;60(4):2102411. doi: 10.1183/13993003.02411-2021. Print 2022 Oct. Eur Respir J. 2022. PMID: 35301248 Free PMC article.
-
Periodic Boosters of COVID-19 Vaccines Do Not Affect the Safety and Efficacy of Immune Checkpoint Inhibitors for Advanced Non-Small Cell Lung Cancer: A Longitudinal Analysis of the Vax-On-Third Study.Cancers (Basel). 2025 Jun 11;17(12):1948. doi: 10.3390/cancers17121948. Cancers (Basel). 2025. PMID: 40563598 Free PMC article.
-
Association of immune-related adverse events with COVID-19 pneumonia in lung cancer patients receiving immune checkpoint inhibitors: a cross-sectional study in China.BMC Cancer. 2023 Nov 6;23(1):1069. doi: 10.1186/s12885-023-11584-w. BMC Cancer. 2023. PMID: 37932685 Free PMC article.
-
Predicting survival in patients with SARS-CoV-2 based on cytokines and soluble immune checkpoint regulators.Front Cell Infect Microbiol. 2024 Nov 25;14:1397297. doi: 10.3389/fcimb.2024.1397297. eCollection 2024. Front Cell Infect Microbiol. 2024. PMID: 39654974 Free PMC article.
References
-
- Awadasseid A., Wu Y., Tanaka Y., Zhang W. Initial success in the identification and management of the coronavirus disease 2019 (COVID-19) indicates human-to-human transmission in Wuhan, China. Int. J. Biol. Sci. 2020;16:1846–1860. http://www.ijbs.com/v1816p1846.htm - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

