Roars, groans and moans: Anatomical correlates of vocal diversity in polygynous deer
- PMID: 34342877
- PMCID: PMC8602020
- DOI: 10.1111/joa.13519
Roars, groans and moans: Anatomical correlates of vocal diversity in polygynous deer
Abstract
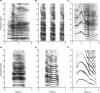
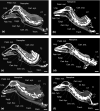
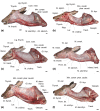
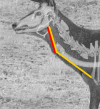
Eurasian deer are characterized by the extraordinary diversity of their vocal repertoires. Male sexual calls range from roars with relatively low fundamental frequency (hereafter fo ) in red deer Cervus elaphus, to moans with extremely high fo in sika deer Cervus nippon, and almost infrasonic groans with exceptionally low fo in fallow deer Dama dama. Moreover, while both red and fallow males are capable of lowering their formant frequencies during their calls, sika males appear to lack this ability. Female contact calls are also characterized by relatively less pronounced, yet strong interspecific differences. The aim of this study is to examine the anatomical bases of these inter-specific and inter-sexual differences by identifying if the acoustic variation is reflected in corresponding anatomical variation. To do this, we investigated the vocal anatomy of male and female specimens of each of these three species. Across species and sexes, we find that the observed acoustic variability is indeed related to expected corresponding anatomical differences, based on the source-filter theory of vocal production. At the source level, low fo is associated with larger vocal folds, whereas high fo is associated with smaller vocal folds: sika deer have the smallest vocal folds and male fallow deer the largest. Red and sika deer vocal folds do not appear to be sexually dimorphic, while fallow deer exhibit strong sexual dimorphism (after correcting for body size differences). At the filter level, the variability in formants is related to the configuration of the vocal tract: in fallow and red deer, both sexes have evolved a permanently descended larynx (with a resting position of the larynx much lower in males than in females). Both sexes also have the potential for momentary, call-synchronous vocal tract elongation, again more pronounced in males than in females. In contrast, the resting position of the larynx is high in both sexes of sika deer and the potential for further active vocal tract elongation is virtually absent in both sexes. Anatomical evidence suggests an evolutionary reversal in larynx position within sika deer, that is, a secondary larynx ascent. Together, our observations confirm that the observed diversity of vocal behaviour in polygynous deer is supported by strong anatomical differences, highlighting the importance of anatomical specializations in shaping mammalian vocal repertoires. Sexual selection is discussed as a potential evolutionary driver of the observed vocal diversity and sexual dimorphisms.
Keywords: acoustic variation; descended larynx; fallow deer; female contact calls; male sexual calls; polygynous deer; red deer; sexual dimorphism; sexual selection; sika deer; source-filter theory; vocal anatomy; vocal production; vocal repertoire.
© 2021 The Authors. Journal of Anatomy published by John Wiley & Sons Ltd on behalf of Anatomical Society.
Figures










References
-
- Apollonio, M. , Festa‐Bianchet, M. , Mari, F. , Mattioli, S. & Sarno, B. (1992) To lek or not to lek: mating strategies of male fallow deer. Behavioral Ecology, 3, 25–31.
-
- Azorit, C. , Analla, M. & Muñoz‐Cobo, J. (2003) Variation of mandible size in red deer Cervus elaphus hispanicus from southern Spain. Acta Theriologica, 48, 221–228.
-
- Berthommier, F. , Boë, L.‐J. , Meguerditchian, A. , Sawallis, T.R. & Captier, G. (2017). Comparative anatomy of the baboon and human vocal tracts: renewal of methods, data, and hypotheses. In: Boë, L.‐J. , Fagot, J. , Perrier, P. & Schwartz, J.‐L. (Eds.) Origins of human language: continuities and discontinuities with nonhuman primates, pp. 101–135. Speech production and perception, Berlin: Peter Lang GmbH, Internationaler Verlag der Wissenschaften. 78‐3‐631‐73726‐2. 10.3726/b12405. hal‐01649499v2. - DOI
-
- Blant, M. & Gaillard, J.M. (2004) Use of biometric body variables as indicators of roe deer (Capreolus capreolus) population density changes. Game and Wildlife Science, 21, 21–40.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous

