Characterization and allogeneic transplantation of a novel transgenic cone-rich donor mouse line
- PMID: 34343570
- PMCID: PMC8429259
- DOI: 10.1016/j.exer.2021.108715
Characterization and allogeneic transplantation of a novel transgenic cone-rich donor mouse line
Abstract
Objectives: Cone photoreceptor transplantation is a potential treatment for macular diseases. The optimal conditions for cone transplantation are poorly understood, partly because of the scarcity of cones in donor mice. To facilitate allogeneic cone photoreceptor transplantation studies in mice, we aimed to create and characterize a donor mouse model containing a cone-rich retina with a cone-specific enhanced green fluorescent protein (EGFP) reporter.
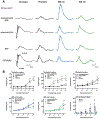
Methods: We generated OPN1LW-EGFP/NRL-/- mice by crossing NRL-/- and OPN1LW-EGFP mice. We characterized the anatomical phenotype of OPN1LW-EGFP/NRL-/- mice using multimodal confocal scanning laser ophthalmoscopy (cSLO) imaging, immunohistology, and transmission electron microscopy. We evaluated retinal function using electroretinography (ERG), including 465 and 525 nm chromatic stimuli. Retinal sheets and cell suspensions from OPN1LW-EGFP/NRL-/- mice were transplanted subretinally into immunodeficient Rd1 mice.
Results: OPN1LW-EGFP/NRL-/- retinas were enriched with OPN1LW-EGFP+ and S-opsin+ cone photoreceptors in a dorsal-ventral distribution gradient. Cone photoreceptors co-expressing OPNL1W-EGFP and S-opsin significantly increased in OPN1LW-EGFP/NRL-/- compared to OPN1LW-EGFP mice. Temporal dynamics of rosette formation in the OPN1LW-EGFP/NRL-/- were similar as the NRL-/- with peak formation at P15. Rosettes formed preferentially in the ventral retina. The outer retina in P35 OPN1LW-EGFP/NRL-/- was thinner than NRL-/- controls. The OPN1LW-EGFP/NRL-/- ERG response amplitudes to 465 nm stimulation were similar to, but to 535 nm stimulation were lower than, NRL-/- controls. Three months after transplantation, the suspension grafts showed greater macroscopic degradation than sheet grafts. Retinal sheet grafts from OPN1LW-EGFP/NRL-/- mice showed greater S-opsin + cone survival than suspension grafts from the same strain.
Conclusions: OPN1LW-EGFP/NRL-/- retinae were enriched with S-opsin+ photoreceptors. Sustained expression of EGFP facilitated the longitudinal tracking of transplanted donor cells. Transplantation of cone-rich retinal grafts harvested prior to peak rosette formation survived and differentiated into cone photoreceptor subtypes. Photoreceptor sheet transplantation may promote greater macroscopic graft integrity and S-opsin+ cone survival than cell suspension transplantation, although the mechanism underlying this observation is unclear at present. This novel cone-rich reporter mouse strain may be useful to study the influence of graft structure on cone survival.
Keywords: Age-related macular degeneration; Cone photoreceptor; Cone transplantation; Degenerative retinal diseases; Optic coherence tomography; Stem cell therapy.
Copyright © 2021 The Authors. Published by Elsevier Ltd.. All rights reserved.
Conflict of interest statement
Declarations of interest:
None
Figures




References
-
- Akimoto M, Cheng H, Zhu D, Brzezinski JA, Khanna R, Filippova E, Oh EC, Jing Y, Linares JL, Brooks M, Zareparsi S, Mears AJ, Hero A, Glaser T, Swaroop A, 2006. Targeting of GFP to newborn rods by Nrl promoter and temporal expression profiling of flow-sorted photoreceptors. Proc Natl Acad Sci U S A 103, 3890–3895. - PMC - PubMed
-
- Anderson DH, Guerin CJ, Erickson PA, Stern WH, Fisher SK, 1986. Morphological recovery in the reattached retina. Invest Ophthalmol Vis Sci 27, 168–183. - PubMed
-
- Applebury ML, Antoch MP, Baxter LC, Chun LL, Falk JD, Farhangfar F, Kage K, Krzystolik MG, Lyass LA, Robbins JT, 2000. The murine cone photoreceptor: a single cone type expresses both S and M opsins with retinal spatial patterning. Neuron 27, 513–523. - PubMed
-
- Arden G, Wolf J, Berninger T, Hogg CR, Tzekov R, Holder GE, 1999. S-cone ERGs elicited by a simple technique in normals and in tritanopes. Vision Res 39, 641–650. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

