Update from the European Committee on Antimicrobial Susceptibility Testing (EUCAST)
- PMID: 34346716
- PMCID: PMC8925892
- DOI: 10.1128/JCM.00276-21
Update from the European Committee on Antimicrobial Susceptibility Testing (EUCAST)
Abstract
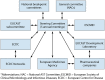
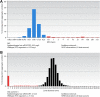
The European Committee on Antimicrobial Susceptibility Testing (EUCAST) is an international susceptibility testing committee, organized by the European Society for Clinical Microbiology and Infectious Diseases (ESCMID) and functioning as the breakpoint advisory committee of the European Medicines Agency (EMA). The original remit of EUCAST was to harmonize European clinical breakpoints, but very soon, the activities expanded beyond the borders of Europe and included newly licensed agents in Europe. Among the milestones were the aggregating of large numbers of MIC distributions, creating software to display these distributions, the EUCAST concept of identifying epidemiological cutoff values (ECOFF), and the development of a EUCAST disk diffusion method. The EUCAST Development Laboratory has played a critical role in the development of antimicrobial susceptibility testing (AST) methodology, including development work for novel antimicrobial agents and for rapid AST directly from blood culture bottles. EUCAST has several standing subcommittees, including for AST in fungi (AFST) and mycobacteria (AMST) and for microorganisms of veterinary interest (VetCAST), and ad hoc subcommittees on subjects such as anaerobic bacteria, MIC and zone diameter distributions and epidemiological cutoff values, the relationship between phenotypic and genotypic resistance, and expert rules and methods for the detection of resistance mechanisms. All EUCAST decisions are subjected to the EUCAST public consultation process, the only exception being breakpoints of novel antimicrobial agents where confidentiality agreements during the licensing process prevent public participation. EUCAST has recently revised the definitions of clinical susceptibility interpretive categories S, I, and R, acknowledging the intimate relationship between drug exposure and susceptibility reporting.
Keywords: antifungal; antimicrobial susceptibility testing; breakpoint; drug development; minimal inhibitory concentration; mycobacteria; pharmacokinetics and pharmacodynamics; resistance mechanisms; veterinary.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Brown D, Cantón R, Dubreuil L, Gatermann S, Giske C, MacGowan A, Martínez-Martínez L, Mouton J, Skov R, Steinbakk M, Walton C, Heuer O, Struelens MJ, Diaz Högberg L, Kahlmeter G. 2015. Widespread implementation of EUCAST breakpoints for antibacterial susceptibility testing in Europe. Eurosurveillance 20:21008. 10.2807/1560-7917.ES2015.20.2.21008. - DOI - PubMed
-
- European Committee on Antimicrobial Susceptibility Testing. EUCAST general website. www.eucast.org.
-
- European Committee on Antimicrobial Susceptibility Testing. EUCAST public consultations. https://www.eucast.org/publications_and_documents/consultations/.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

