Porous functionalized polymers enable generating and transporting hyperpolarized mixtures of metabolites
- PMID: 34349114
- PMCID: PMC8338986
- DOI: 10.1038/s41467-021-24279-2
Porous functionalized polymers enable generating and transporting hyperpolarized mixtures of metabolites
Abstract
Hyperpolarization by dissolution dynamic nuclear polarization (dDNP) has enabled promising applications in spectroscopy and imaging, but remains poorly widespread due to experimental complexity. Broad democratization of dDNP could be realized by remote preparation and distribution of hyperpolarized samples from dedicated facilities. Here we show the synthesis of hyperpolarizing polymers (HYPOPs) that can generate radical- and contaminant-free hyperpolarized samples within minutes with lifetimes exceeding hours in the solid state. HYPOPs feature tunable macroporous porosity, with porous volumes up to 80% and concentration of nitroxide radicals grafted in the bulk matrix up to 285 μmol g-1. Analytes can be efficiently impregnated as aqueous/alcoholic solutions and hyperpolarized up to P(13C) = 25% within 8 min, through the combination of 1H spin diffusion and 1H → 13C cross polarization. Solutions of 13C-analytes of biological interest hyperpolarized in HYPOPs display a very long solid-state 13C relaxation times of 5.7 h at 3.8 K, thus prefiguring transportation over long distances.
© 2021. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
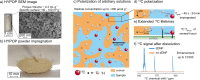


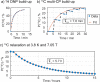

Similar articles
-
Dynamic Nuclear Polarization with Conductive Polymers.Angew Chem Int Ed Engl. 2024 Dec 2;63(49):e202409510. doi: 10.1002/anie.202409510. Epub 2024 Oct 31. Angew Chem Int Ed Engl. 2024. PMID: 39264818
-
Full optimization of dynamic nuclear polarization on a 1 tesla benchtop polarizer with hyperpolarizing solids.Phys Chem Chem Phys. 2024 Aug 22;26(33):22049-22061. doi: 10.1039/d4cp02022g. Phys Chem Chem Phys. 2024. PMID: 39114945 Free PMC article.
-
Hyperpolarized water through dissolution dynamic nuclear polarization with UV-generated radicals.Commun Chem. 2020 May 8;3(1):57. doi: 10.1038/s42004-020-0301-6. Commun Chem. 2020. PMID: 36703471 Free PMC article.
-
Use of dissolved hyperpolarized species in NMR: Practical considerations.Prog Nucl Magn Reson Spectrosc. 2020 Jun-Aug;118-119:74-90. doi: 10.1016/j.pnmrs.2020.03.002. Epub 2020 Apr 28. Prog Nucl Magn Reson Spectrosc. 2020. PMID: 32883450 Review.
-
Hyperpolarization via dissolution dynamic nuclear polarization: new technological and methodological advances.MAGMA. 2021 Feb;34(1):5-23. doi: 10.1007/s10334-020-00894-w. Epub 2020 Nov 13. MAGMA. 2021. PMID: 33185800 Review.
Cited by
-
Signal enhancement of hyperpolarized 15 N sites in solution-increase in solid-state polarization at 3.35 T and prolongation of relaxation in deuterated water mixtures.NMR Biomed. 2022 Nov;35(11):e4787. doi: 10.1002/nbm.4787. Epub 2022 Jul 14. NMR Biomed. 2022. PMID: 35704397 Free PMC article.
-
Fine optimization of a dissolution dynamic nuclear polarization experimental setting for 13C NMR of metabolic samples.Magn Reson (Gott). 2022 Sep 29;3(2):183-202. doi: 10.5194/mr-3-183-2022. eCollection 2022. Magn Reson (Gott). 2022. PMID: 37904870 Free PMC article.
-
Boosting 1H and 13C NMR signals by orders of magnitude on a bench.Sci Adv. 2024 Dec 6;10(49):eadq3780. doi: 10.1126/sciadv.adq3780. Epub 2024 Dec 4. Sci Adv. 2024. PMID: 39630888 Free PMC article.
-
Paramagnetic relaxivity of delocalized long-lived states of protons in chains of CH2 groups.Magn Reson (Gott). 2023 Feb 16;4(1):47-56. doi: 10.5194/mr-4-47-2023. eCollection 2023. Magn Reson (Gott). 2023. PMID: 37904798 Free PMC article.
-
Parallel detection of chemical reactions in a microfluidic platform using hyperpolarized nuclear magnetic resonance.Lab Chip. 2023 Nov 21;23(23):4950-4958. doi: 10.1039/d3lc00474k. Lab Chip. 2023. PMID: 37906028 Free PMC article.
References
-
- Elliott, S. J. et al. Practical dissolution dynamic nuclear polarization, Progress in Nuclear Magnetic Resonance Spectroscopy. (2021) 10.1016/j.pnmrs.2021.04.002. - PubMed
Publication types
LinkOut - more resources
Full Text Sources

