The degree of polymerization and sulfation patterns in heparan sulfate are critical determinants of cytomegalovirus entry into host cells
- PMID: 34352038
- PMCID: PMC8384199
- DOI: 10.1371/journal.ppat.1009803
The degree of polymerization and sulfation patterns in heparan sulfate are critical determinants of cytomegalovirus entry into host cells
Abstract
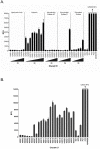
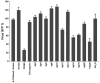
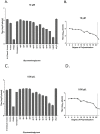
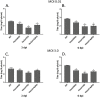
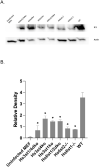
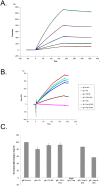
Several enveloped viruses, including herpesviruses attach to host cells by initially interacting with cell surface heparan sulfate (HS) proteoglycans followed by specific coreceptor engagement which culminates in virus-host membrane fusion and virus entry. Interfering with HS-herpesvirus interactions has long been known to result in significant reduction in virus infectivity indicating that HS play important roles in initiating virus entry. In this study, we provide a series of evidence to prove that specific sulfations as well as the degree of polymerization (dp) of HS govern human cytomegalovirus (CMV) binding and infection. First, purified CMV extracellular virions preferentially bind to sulfated longer chain HS on a glycoarray compared to a variety of unsulfated glycosaminoglycans including unsulfated shorter chain HS. Second, the fraction of glycosaminoglycans (GAG) displaying higher dp and sulfation has a larger impact on CMV titers compared to other fractions. Third, cell lines deficient in specific glucosaminyl sulfotransferases produce significantly reduced CMV titers compared to wild-type cells and virus entry is compromised in these mutant cells. Finally, purified glycoprotein B shows strong binding to heparin, and desulfated heparin analogs compete poorly with heparin for gB binding. Taken together, these results highlight the significance of HS chain length and sulfation patterns in CMV attachment and infectivity.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures