Association between Influenza Vaccination and the Risk of Bell's Palsy in the Korean Elderly
- PMID: 34358162
- PMCID: PMC8310133
- DOI: 10.3390/vaccines9070746
Association between Influenza Vaccination and the Risk of Bell's Palsy in the Korean Elderly
Abstract
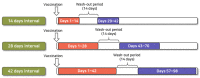
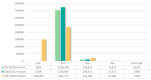
Previous studies have shown controversial results on the risk of Bell's palsy after influenza vaccination. Since the antigenic components of influenza vaccine can vary from season to season, continuous safety monitoring is required. The aim of the present study was to determine whether there was an increased risk of Bell's palsy in the elderly after influenza vaccination between the 2015/2016 and 2017/2018 flu seasons. This study included the elderly who received influenza vaccinations for three flu seasons using a large-linked database of vaccination registration data from the Korea Disease Control and Prevention Agency and the National Health Insurance Service claims data. We used a self-controlled risk interval design with a risk interval of 1 to 42 days and a control interval of 57 to 98 days postvaccination and calculated the incidence rate ratio. To ensure the robustness of the results, sensitivity analyses were also carried out with different risk and control intervals. Of 4,653,440 elderly people who received the influenza vaccine, there was no statistically significant increase in the risk of Bell's palsy (IRR: 0.99, 95% CI: 0.92-1.07). Similar results were found in analysis results for each season and the results of the sensitivity analyses excluding the 2017/2018 season. In conclusion, we found no evidence of an increased risk of Bell's palsy after influenza vaccination. The results of our study provide reassurance about the safety of the influenza vaccine NIP program. However, it is necessary to continuously monitor the risk of Bell's palsy during future flu seasons.
Keywords: Bell’s palsy; adverse events; elderly people; influenza vaccine; large-linked database; self-controlled risk interval design.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Bell's palsy and influenza(H1N1)pdm09 containing vaccines: A self-controlled case series.PLoS One. 2017 May 3;12(5):e0175539. doi: 10.1371/journal.pone.0175539. eCollection 2017. PLoS One. 2017. PMID: 28467420 Free PMC article.
-
Messenger RNA Coronavirus Disease 2019 (COVID-19) Vaccination With BNT162b2 Increased Risk of Bell's Palsy: A Nested Case-Control and Self-Controlled Case Series Study.Clin Infect Dis. 2023 Feb 8;76(3):e291-e298. doi: 10.1093/cid/ciac460. Clin Infect Dis. 2023. PMID: 35675702 Free PMC article.
-
Use of the inactivated intranasal influenza vaccine and the risk of Bell's palsy in Switzerland.N Engl J Med. 2004 Feb 26;350(9):896-903. doi: 10.1056/NEJMoa030595. N Engl J Med. 2004. PMID: 14985487
-
Bell's Palsy and COVID-19 Vaccination: A Systematic Review.Med J Islam Repub Iran. 2022 Jul 30;36:85. doi: 10.47176/mjiri.36.85. eCollection 2022. Med J Islam Repub Iran. 2022. PMID: 36128311 Free PMC article. Review.
-
Recurrent Bell's palsy.Clin Otolaryngol. 2019 May;44(3):305-312. doi: 10.1111/coa.13293. Epub 2019 Feb 22. Clin Otolaryngol. 2019. PMID: 30672667
Cited by
-
Risk of peripheral facial palsy following parenteral inactivated influenza vaccination in the elderly Chinese population.Front Public Health. 2023 Jan 24;11:1047391. doi: 10.3389/fpubh.2023.1047391. eCollection 2023. Front Public Health. 2023. PMID: 36761129 Free PMC article.
-
Facial and Abducens Nerve Palsies Following COVID-19 Vaccination: Report of Two Cases.Neuroophthalmology. 2022 Feb 15;46(3):203-206. doi: 10.1080/01658107.2022.2032204. eCollection 2022. Neuroophthalmology. 2022. PMID: 35574171 Free PMC article.
-
Safety Assessment of Influenza Vaccination for Neurological Outcomes Among Older Adults in Japan: A Self-Controlled Case Series Study.Pharmacoepidemiol Drug Saf. 2025 Jan;34(1):e70082. doi: 10.1002/pds.70082. Pharmacoepidemiol Drug Saf. 2025. PMID: 39777941 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources

