Evaluating the antidiabetic effects of R-verapamil in type 1 and type 2 diabetes mellitus mouse models
- PMID: 34358247
- PMCID: PMC8345870
- DOI: 10.1371/journal.pone.0255405
Evaluating the antidiabetic effects of R-verapamil in type 1 and type 2 diabetes mellitus mouse models
Abstract
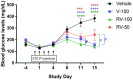
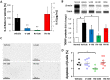
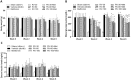
The global incidence of diabetes mellitus (DM) is increasing. Types 1 and 2 DM are associated with declining β-cell function. Verapamil (50% S-verapamil and 50% R-verapamil) can treat DM by downregulating thioredoxin-interacting protein (TXNIP), which induces islet β-cell apoptosis. However, it may also induce cardiovascular side effects as S-verapamil is negatively inotropic. In contrast, R-verapamil only weakly induces adverse cardiac effects. In this study, we aimed to determine the antidiabetic efficacy and cardiovascular safety of R-verapamil. We examined R- and S-verapamil binding through in vitro studies. Streptozotocin-induced type 1 and db/db type 2 DM mouse models were used to assess the antidiabetic efficacy of verapamil. IL-6, blood glucose (BG), Txnip expression, and β-cells were evaluated in streptozotocin-induced diabetic mice, while body weight, BG, and serum insulin were measured in the db/db mice. In the type 1 DM study, 100 mg/kg/day R-verapamil and racemic verapamil lowered BG, downregulated Txnip expression, and reduced β-cell apoptosis. In the type 2 DM study, the optimal R-verapamil dosage was 60 mg/kg/day and it lowered BG and raised serum insulin. However, efficacy did not increase with R-verapamil dosage. R-verapamil combined with metformin/acarbose improved BG and serum insulin more effectively than metformin/acarbose alone or verapamil combined with acarbose. R-verapamil had weaker cardiovascular side effects than S-verapamil. R-verapamil was 9.0× and 3.4× less effective than S-verapamil at inhibiting atrial inotropy and ileal contractility, respectively. It was also 8.7× weaker than S-verapamil as an agonist of somatostatin receptor type 2 (SSTR2), inhibiting ileal neurogenic contraction. Hence, R-verapamil may be an optimal DM treatment as it is safe, improves glycemic control, and preserves β-cell function both as monotherapy and in combination with metformin or acarbose. R-Verapamil has potential for delaying or arresting DM progression and improving patients' quality of life.
Conflict of interest statement
All the authors and Center Laboratories Inc. have not served on the editorial board of PLOS ONE. All the authors and Center Laboratories Inc. have not acted as an expert witness in relevant legal proceedings. All the authors and Center Laboratories Inc. have not sat on a committee for any organization that may benefit from publication of PLOS ONE. This Competing Interests statement does not alter our adherence to PLOS ONE policies on sharing data and materials.
Figures





Similar articles
-
A Novel Resolution of Diabetes: C-C Chemokine Motif Ligand 4 Is a Common Target in Different Types of Diabetes by Protecting Pancreatic Islet Cell and Modulating Inflammation.Front Immunol. 2021 Apr 23;12:650626. doi: 10.3389/fimmu.2021.650626. eCollection 2021. Front Immunol. 2021. PMID: 33968046 Free PMC article.
-
Extract of Ginkgo Biloba Ameliorates Streptozotocin-Induced Type 1 Diabetes Mellitus and High-Fat Diet-Induced Type 2 Diabetes Mellitus in Mice.Int J Med Sci. 2015 Nov 23;12(12):987-94. doi: 10.7150/ijms.13339. eCollection 2015. Int J Med Sci. 2015. PMID: 26664261 Free PMC article.
-
Proposed mechanisms for oligonucleotide IMT504 induced diabetes reversion in a mouse model of immunodependent diabetes.Am J Physiol Endocrinol Metab. 2016 Aug 1;311(2):E380-95. doi: 10.1152/ajpendo.00104.2016. Epub 2016 Jun 21. Am J Physiol Endocrinol Metab. 2016. PMID: 27329801
-
Oral antidiabetic agents: current role in type 2 diabetes mellitus.Drugs. 2005;65(3):385-411. doi: 10.2165/00003495-200565030-00005. Drugs. 2005. PMID: 15669880 Review.
-
Protection of pancreatic beta-cells: is it feasible?Nutr Metab Cardiovasc Dis. 2008 Jan;18(1):74-83. doi: 10.1016/j.numecd.2007.05.004. Nutr Metab Cardiovasc Dis. 2008. PMID: 18096375 Review.
Cited by
-
Automated gait analysis indicates efficacy of T-type calcium channel inhibition for mitigation of disrupted calcium signalling in an SCA5 mouse model.Sci Rep. 2025 Jul 1;15(1):20990. doi: 10.1038/s41598-025-05511-1. Sci Rep. 2025. PMID: 40594196 Free PMC article.
-
A Randomized Controlled Trial of R-Form Verapamil Added to Ongoing Metformin Therapy in Patients with Type 2 Diabetes.J Clin Endocrinol Metab. 2022 Sep 28;107(10):e4063-e4071. doi: 10.1210/clinem/dgac436. J Clin Endocrinol Metab. 2022. PMID: 35917580 Free PMC article. Clinical Trial.
-
Preliminary evaluation of the antiglycoxidant activity of verapamil using various in vitro and in silico biochemical/biophysical methods.Front Pharmacol. 2023 Nov 28;14:1293295. doi: 10.3389/fphar.2023.1293295. eCollection 2023. Front Pharmacol. 2023. PMID: 38089049 Free PMC article.
-
Emerging Anti-Diabetic Drugs for Beta-Cell Protection in Type 1 Diabetes.Cells. 2023 May 25;12(11):1472. doi: 10.3390/cells12111472. Cells. 2023. PMID: 37296593 Free PMC article. Review.
-
Effect of Verapamil on Blood Glucose in Type 1 and Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis.Cardiovasc Drugs Ther. 2025 Mar 20. doi: 10.1007/s10557-025-07683-4. Online ahead of print. Cardiovasc Drugs Ther. 2025. PMID: 40111679 Review.
References
-
- Centers for Disease Control and Prevention C. National diabetes statistics report, 2020. Atlanta, GA: Centers for Disease Control and Prevention, US Department of Health and Human Services. 2020.
-
- Cho NH, Kirigia J, Mbanya JC, Ogurstova K, Guariguata L, Rathmann W. IDF DIABETES ATLAS—8TH EDITION. The International Diabetes Federation; 2017.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

