Autophagy modulates endothelial junctions to restrain neutrophil diapedesis during inflammation
- PMID: 34363750
- PMCID: PMC8459396
- DOI: 10.1016/j.immuni.2021.07.012
Autophagy modulates endothelial junctions to restrain neutrophil diapedesis during inflammation
Abstract
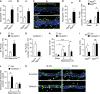
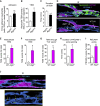
The migration of neutrophils from the blood circulation to sites of infection or injury is a key immune response and requires the breaching of endothelial cells (ECs) that line the inner aspect of blood vessels. Unregulated neutrophil transendothelial cell migration (TEM) is pathogenic, but the molecular basis of its physiological termination remains unknown. Here, we demonstrated that ECs of venules in inflamed tissues exhibited a robust autophagic response that was aligned temporally with the peak of neutrophil trafficking and was strictly localized to EC contacts. Genetic ablation of EC autophagy led to excessive neutrophil TEM and uncontrolled leukocyte migration in murine inflammatory models, while pharmacological induction of autophagy suppressed neutrophil infiltration into tissues. Mechanistically, autophagy regulated the remodeling of EC junctions and expression of key EC adhesion molecules, facilitating their intracellular trafficking and degradation. Collectively, we have identified autophagy as a modulator of EC leukocyte trafficking machinery aimed at terminating physiological inflammation.
Keywords: ATG16L1; ATG5; PECAM-1; autophagy; diapedesis; endothelium; extravasation; inflammation; junctions; neutrophils.
Copyright © 2021 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no competing interests.
Figures






References
-
- Alva J.A., Zovein A.C., Monvoisin A., Murphy T., Salazar A., Harvey N.L., Carmeliet P., Iruela-Arispe M.L. VE-Cadherin-Cre-recombinase transgenic mouse: a tool for lineage analysis and gene deletion in endothelial cells. Dev. Dyn. 2006;235:759–767. - PubMed
-
- Arganda-Carreras I., Sorzano C., Marabini R., Carazo J., Ortiz-de-Solorzano C., Kybic J. In: Computer Vision Approaches to Medical Image Analysis. Beichel R.R., Sonka M., editors. Springer; 2006. Consistent and Elastic Registration of Histological Sections Using Vector-Spline Regularization.
-
- Bentham J., Morris D.L., Graham D.S.C., Pinder C.L., Tombleson P., Behrens T.W., Martín J., Fairfax B.P., Knight J.C., Chen L. Genetic association analyses implicate aberrant regulation of innate and adaptive immunity genes in the pathogenesis of systemic lupus erythematosus. Nat. Genet. 2015;47:1457–1464. - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

