Mechanistic Modeling of a Novel Oncolytic Virus, V937, to Describe Viral Kinetic and Dynamic Processes Following Intratumoral and Intravenous Administration
- PMID: 34366859
- PMCID: PMC8343024
- DOI: 10.3389/fphar.2021.705443
Mechanistic Modeling of a Novel Oncolytic Virus, V937, to Describe Viral Kinetic and Dynamic Processes Following Intratumoral and Intravenous Administration
Abstract
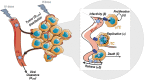
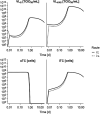
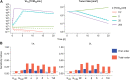
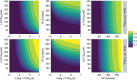
V937 is an investigational novel oncolytic non-genetically modified Kuykendall strain of Coxsackievirus A21 which is in clinical development for the treatment of advanced solid tumor malignancies. V937 infects and lyses tumor cells expressing the intercellular adhesion molecule I (ICAM-I) receptor. We integrated in vitro and in vivo data from six different preclinical studies to build a mechanistic model that allowed a quantitative analysis of the biological processes of V937 viral kinetics and dynamics, viral distribution to tumor, and anti-tumor response elicited by V937 in human xenograft models in immunodeficient mice following intratumoral and intravenous administration. Estimates of viral infection and replication which were calculated from in vitro experiments were successfully used to describe the tumor response in vivo under various experimental conditions. Despite the predicted high clearance rate of V937 in systemic circulation (t1/2 = 4.3 min), high viral replication was observed in immunodeficient mice which resulted in tumor shrinkage with both intratumoral and intravenous administration. The described framework represents a step towards the quantitative characterization of viral distribution, replication, and oncolytic effect of a novel oncolytic virus following intratumoral and intravenous administrations in the absence of an immune response. This model may further be expanded to integrate the role of the immune system on viral and tumor dynamics to support the clinical development of oncolytic viruses.
Keywords: mechanistic modeling; oncolytic virus; tumor distribution; viral dynamics; viral kinetics.
Copyright © 2021 Parra-Guillen, Freshwater, Cao, Mayawala, Zalba, Garrido, de Alwis and Troconiz.
Conflict of interest statement
Authors TF, YC, KM, and DA were employed by the company Merck & Co, Inc, Kenilworth, United States. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Al-Tuwairqi S. M., Al-Johani N. O., Simbawa E. A. (2020). Modeling Dynamics of Cancer Virotherapy with Immune Response. Adv. Differ. Equ 2020. 10.1186/s13662-020-02893-6 - DOI
LinkOut - more resources
Full Text Sources

