Potentiation of Acetylcholine-Induced Relaxation of Aorta in Male UC Davis Type 2 Diabetes Mellitus (UCD-T2DM) Rats: Sex-Specific Responses
- PMID: 34366875
- PMCID: PMC8339592
- DOI: 10.3389/fphys.2021.616317
Potentiation of Acetylcholine-Induced Relaxation of Aorta in Male UC Davis Type 2 Diabetes Mellitus (UCD-T2DM) Rats: Sex-Specific Responses
Abstract
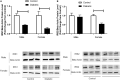
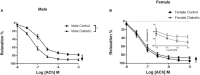
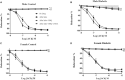
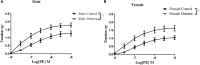
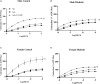
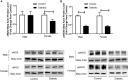
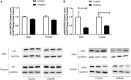
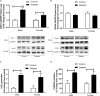
Previous reports suggest that diabetes may differentially affect the vascular beds of females and males. The objectives of this study were to examine whether there were (1) sex differences in aortic function and (2) alterations in the relative contribution of endothelium-derived relaxing factors in modulating aortic reactivity in UC Davis Type 2 Diabetes Mellitus (UCD-T2DM) rats. Endothelium-dependent vasorelaxation (EDV) in response to acetylcholine (ACh) was measured in aortic rings before and after exposure to pharmacological inhibitors. Relaxation responses to sodium nitroprusside were assessed in endothelium-denuded rings. Moreover, contractile responses to phenylephrine (PE) were measured before and after incubation of aortic rings with a nitric oxide synthase (NOS) inhibitor in the presence of indomethacin. Metabolic parameters and expression of molecules associated with vascular and insulin signaling as well as reactive oxygen species generation were determined. Diabetes slightly but significantly impaired EDV in response to ACh in aortas from females but potentiated the relaxation response in males. The potentiation of EDV in diabetic male aortas was accompanied by a traces of nitric oxide (NO)- and prostanoid-independent relaxation and elevated aortic expression of small- and intermediate conductance Ca2+-activated K+ channels in this group. The smooth muscle sensitivity to NO was not altered, whereas the responsiveness to PE was significantly enhanced in aortas of diabetic groups in both sexes. Endothelium-derived NO during smooth muscle contraction, as assessed by the potentiation of the response to PE after NOS inhibition, was reduced in aortas of diabetic rats regardless of sex. Accordingly, decreases in pAkt and peNOS were observed in aortas from diabetic rats in both sexes compared with controls. Our data suggest that a decrease in insulin sensitivity via pAkt-peNOS-dependent signaling and an increase in oxidative stress may contribute to the elevated contractile responses observed in diabetic aortas in both sexes. This study demonstrates that aortic function in UCD-T2DM rats is altered in both sexes. Here, we provide the first evidence of sexual dimorphism in aortic relaxation in UCD-T2DM rats.
Keywords: aorta; insulin resistance; nitric oxide; sex differences; type-2 diabetes.
Copyright © 2021 Akther, Razan, Shaligram, Graham, Stanhope, Allen, Vázquez-Medina, Havel and Rahimian.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
-
- Abebe W., Harris K. H., MacLeod K. M. (1990). Enhanced contractile responses of arteries from diabetic rats to α1-adrenoceptor stimulation in the absence and presence of extracellular calcium. J. Cardiovasc. Pharmacol. 16 239–248. - PubMed
-
- Barrett-Connor E. (1994). Heart disease in women. Fertil. Steril. 62 127S–132S. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

