Comparison between Intra-Articular Injection of Infrapatellar Fat Pad (IPFP) Cell Concentrates and IPFP-Mesenchymal Stem Cells (MSCs) for Cartilage Defect Repair of the Knee Joint in Rabbits
- PMID: 34367294
- PMCID: PMC8337123
- DOI: 10.1155/2021/9966966
Comparison between Intra-Articular Injection of Infrapatellar Fat Pad (IPFP) Cell Concentrates and IPFP-Mesenchymal Stem Cells (MSCs) for Cartilage Defect Repair of the Knee Joint in Rabbits
Abstract
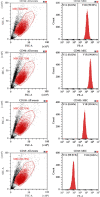
Mesenchymal stem cells (MSCs) have emerged as a promising therapeutic method in regenerative medicine. Our previous research adopted a simple nonenzymatic strategy for the preparation of a new type of ready-to-use infrapatellar fat pad (IPFP) cell concentrates. The aim of this study was to compare the therapeutic efficacy of intra-articular (IA) injection of autologous IPFP cell concentrates and allogeneic IPFP-MSCs obtained from these concentrates in a rabbit articular cartilage defect model. IPFP-MSCs sprouting from the IPFP cell concentrates were characterized via flow cytometry as well as based on their potential for differentiation into adipocytes, osteoblasts, and chondrocytes. In the rabbit model, cartilage defects were created on the trochlear groove, followed by treatment with IPFP cell concentrates, IPFP-MSCs, or normal saline IA injection. Distal femur samples were evaluated at 6 and 12 weeks posttreatment via macroscopic observation and histological assessment based on the International Cartilage Repair Society (ICRS) macroscopic scoring system as well as the ICRS visual histological assessment scale. The macroscopic score and histological score were significantly higher in the IPFP-MSC group compared to the IPFP cell concentrate group at 12 weeks. Further, both treatment groups had higher scores compared to the normal saline group. In comparison to the latter, the groups treated with IPFP-MSCs and IPFP cell concentrates showed considerably better cartilage regeneration. Overall, IPFP-MSCs represent an effective therapeutic strategy for stimulating articular cartilage regeneration. Further, due to the simple, cost-effective, nonenzymatic, and safe preparation process, IPFP cell concentrates may represent an effective alternative to stem cell-based therapy in the clinic.
Copyright © 2021 Yaguang Han et al.
Conflict of interest statement
The authors declare that there is no conflict of interest regarding the publication of this paper.
Figures




References
LinkOut - more resources
Full Text Sources

