Effects on Growth of Smell and Taste of Milk During Tube Feeding of Preterm Infants: A Randomized Clinical Trial
- PMID: 34369975
- PMCID: PMC8353575
- DOI: 10.1001/jamapediatrics.2021.2336
Effects on Growth of Smell and Taste of Milk During Tube Feeding of Preterm Infants: A Randomized Clinical Trial
Abstract
Importance: Smell and taste of food increase food anticipation, activate gut motility, and stimulate digestion and metabolism. Despite poor growth of many preterm infants in neonatal intensive care units, the smell and taste of milk with tube feeding are not generally considered a regular component of care.
Objective: To determine the effect of smell and taste of milk with tube feeding on weight z scores at discharge from the hospital.
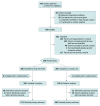
Design, setting, and participants: A randomized, controlled, nonblinded, superiority trial was conducted at 2 perinatal centers between May 9, 2017, and February 1, 2020. Eligible infants (n = 659) were born at less than 29 weeks' postmenstrual age (PMA) and/or with a birth weight of less than 1250 g.
Interventions: Infants were randomly assigned to receive either the smell and taste of milk with each tube feeding or routine care without the provision of smell and taste of milk.
Main outcomes and measures: The primary outcome was weight z score at discharge from any hospital. Secondary outcomes included anthropometric measures at predefined time points, time to full enteral feeds, and other health outcomes associated with prematurity.
Results: Of the 658 infants, a total of 396 infants were randomized; some parents had not been approached for consent (n = 144) or declined participation (n = 117), and 1 infant with consent was not randomized. Of the 396 infants, 196 were assigned to the treatment group (51% male; mean [SD] PMA at birth, 27.5 [2.2] weeks) and 200 were assigned to the control group (52% male; mean [SD] PMA at birth, 27.6 (2.3) weeks). Mean weight z scores at discharge were -0.87 (95% CI, -1.02 to -0.72) for the treatment group and -0.97 (95% CI, -1.11 to -0.83) for the control group (P = .40). The mean difference in z scores between the treatment and control groups at 36 weeks' PMA was 0.21 (95% CI, 0.01 to 0.4; P = .04) for head circumference and 0.26 (95% CI, 0.05 to 0.51; P = .04) for length. There were no clinically notable differences between the study groups for any other anthropometric, feeding, or health outcomes.
Conclusions and relevance: In this randomized clinical trial, regular smell and taste of milk included with tube feeding did not improve weight at discharge in preterm infants. Secondary outcomes suggest exposure to smell and taste may improve head circumference and length at 36 weeks' PMA, but not at discharge. Regular exposure to the smell and taste of milk is a simple and inexpensive intervention with potential benefits and no apparent adverse effects.
Trial registration: anzctr.org.au Identifier: ACTRN12617000583347.
Conflict of interest statement
Figures


References
-
- Stoll BJ, Hansen NI, Bell EF, et al. ; Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network . Trends in care practices, morbidity, and mortality of extremely preterm neonates, 1993-2012. JAMA. 2015;314(10):1039-1051. doi: 10.1001/jama.2015.10244 - DOI - PMC - PubMed
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Medical

