Establishment of a Three-Dimensional In Vitro Model of Equine Papillomavirus Type 2 Infection
- PMID: 34372610
- PMCID: PMC8310375
- DOI: 10.3390/v13071404
Establishment of a Three-Dimensional In Vitro Model of Equine Papillomavirus Type 2 Infection
Abstract
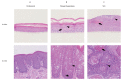
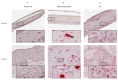
There is growing evidence that equine papillomavirus type 2 (EcPV2) infection is etiologically associated with the development of genital squamous cell carcinoma (SCC) and precursor lesions in equids. However, the precise mechanisms underlying neoplastic progression remain unknown. To allow the study of EcPV2-induced carcinogenesis, we aimed to establish a primary equine cell culture model of EcPV2 infection. Three-dimensional (3D) raft cultures were generated from equine penile perilesional skin, plaques and SCCs. Using histological, molecular biological and immunohistochemical methods, rafts versus corresponding natural tissue sections were compared with regard to morphology, presence of EcPV2 DNA, presence and location of EcPV2 gene transcripts and expression of epithelial, mesenchymal and tumor/proliferation markers. Raft cultures from perilesional skin harboring only a few EcPV2-positive (EcPV2+) cells accurately recapitulated the differentiation process of normal skin, whilst rafts from EcPV2+ penile plaques were structurally organized but showed early hyperplasia. Rafts from EcPV2+ SCCs exhibited pronounced hyperplasia and marked dysplasia. Raft levels of EcPV2 oncogene transcription (E6/E7) and expression of tumor/proliferation markers p53, Ki67 and MCM7 expression positively correlated with neoplastic progression, again reflecting the natural situation. Three-dimensional raft cultures accurately reflected major features of corresponding ex vivo material, thus constituting a valuable new research model to study EcPV2-induced carcinogenesis.
Keywords: 3D model; EcPV2; horse; hyperplasia; papillomavirus; plaque; raft culture; skin; squamous cell carcinoma.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures


Similar articles
-
Papillomavirus-like Particles in Equine Medicine.Viruses. 2023 Jan 25;15(2):345. doi: 10.3390/v15020345. Viruses. 2023. PMID: 36851559 Free PMC article. Review.
-
Paving the way for more precise diagnosis of EcPV2-associated equine penile lesions.BMC Vet Res. 2019 Oct 22;15(1):356. doi: 10.1186/s12917-019-2097-0. BMC Vet Res. 2019. PMID: 31640696 Free PMC article.
-
Equus caballus papillomavirus type 2 (EcPV2)-associated benign penile lesions and squamous cell carcinomas.Vet Med Sci. 2024 Jan;10(1):e1342. doi: 10.1002/vms3.1342. Vet Med Sci. 2024. PMID: 38227707 Free PMC article.
-
EcPV2 DNA in equine squamous cell carcinomas and normal genital and ocular mucosa.Vet Microbiol. 2011 Jan 27;147(3-4):292-9. doi: 10.1016/j.vetmic.2010.07.008. Epub 2010 Jul 22. Vet Microbiol. 2011. PMID: 20813468
-
Papillomavirus infection and squamous cell carcinoma in horses.Vet J. 2017 May;223:48-54. doi: 10.1016/j.tvjl.2017.05.007. Epub 2017 May 31. Vet J. 2017. PMID: 28671071 Review.
Cited by
-
Papillomavirus-like Particles in Equine Medicine.Viruses. 2023 Jan 25;15(2):345. doi: 10.3390/v15020345. Viruses. 2023. PMID: 36851559 Free PMC article. Review.
-
Tumor Cell Plasticity in Equine Papillomavirus-Positive Versus-Negative Squamous Cell Carcinoma of the Head and Neck.Pathogens. 2022 Feb 18;11(2):266. doi: 10.3390/pathogens11020266. Pathogens. 2022. PMID: 35215208 Free PMC article.
References
-
- Campo M.S. Papillomavirus Research: From Natural History to Vaccines and Beyond. 1st ed. Caister Academic Press; Norfolk, UK: 2006. Introduction; pp. 1–2.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

