Quaternary Interaction of the HIV-1 Envelope Trimer with CD4 and Neutralizing Antibodies
- PMID: 34372611
- PMCID: PMC8310203
- DOI: 10.3390/v13071405
Quaternary Interaction of the HIV-1 Envelope Trimer with CD4 and Neutralizing Antibodies
Abstract
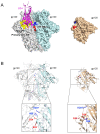
The entry of HIV-1 into host cells is initiated by the interaction of the viral envelope (Env) spike with the CD4 receptor. During this process, the spike undergoes a series of conformational changes that eventually lead to the exposure of the fusion peptide located at the N-terminus of the transmembrane glycoprotein, gp41. Recent structural and functional studies have provided important insights into the interaction of Env with CD4 at various stages. However, a fine elucidation of the earliest events of CD4 contact and its immediate effect on the Env conformation remains a challenge for investigation. Here, we summarize the discovery of the quaternary nature of the CD4-binding site in the HIV-1 Env and the role of quaternary contact in the functional interaction with the CD4 receptor. We propose two models for this initial contact based on the current knowledge and discuss how a better understanding of the quaternary interaction may lead to improved immunogens and antibodies targeting the CD4-binding site.
Keywords: CD4; HIV-1; neutralizing antibodies; quaternary interaction; receptor binding; viral entry.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

