Targeting iron metabolism in cancer therapy
- PMID: 34373750
- PMCID: PMC8344014
- DOI: 10.7150/thno.59092
Targeting iron metabolism in cancer therapy
Abstract
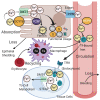
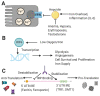
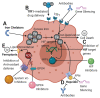
Iron is a critical component of many cellular functions including DNA replication and repair, and it is essential for cell vitality. As an essential element, iron is critical for maintaining human health. However, excess iron can be highly toxic, resulting in oxidative DNA damage. Many studies have observed significant associations between iron and cancer, and the association appears to be more than just coincidental. The chief characteristic of cancers, hyper-proliferation, makes them even more dependent on iron than normal cells. Cancer therapeutics are becoming as diverse as the disease itself. Targeting iron metabolism in cancer cells is an emerging, formidable field of therapeutics. It is a strategy that is highly diverse with regard to specific targets and the various ways to reach them. This review will discuss the importance of iron metabolism in cancer and highlight the ways in which it is being explored as the medicine of tomorrow.
Keywords: Cancer; Chelation; Ferroptosis; Iron metabolism; Therapy.
© The author(s).
Conflict of interest statement
Competing Interests: The authors have declared that no competing interest exists.
Figures
References
-
- Eid R, Arab NT, Greenwood MT. Iron mediated toxicity and programmed cell death: A review and a re-examination of existing paradigms. Biochim Biophys Acta Mol Cell Res. 2017;1864(2):399–430. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

