Reactions of Sodium Diisopropylamide: Liquid-Phase and Solid-Liquid Phase-Transfer Catalysis by N, N, N', N″, N″-Pentamethyldiethylenetriamine
- PMID: 34375095
- PMCID: PMC10042303
- DOI: 10.1021/jacs.1c06528
Reactions of Sodium Diisopropylamide: Liquid-Phase and Solid-Liquid Phase-Transfer Catalysis by N, N, N', N″, N″-Pentamethyldiethylenetriamine
Abstract
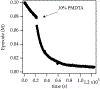
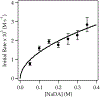
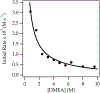
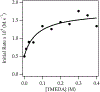
Sodium diisopropylamide (NaDA) in N,N-dimethylethylamine (DMEA) and DMEA-hydrocarbon mixtures with added N,N,N',N″,N″-pentamethyldiethylenetriamine (PMDTA) reacts with alkyl halides, epoxides, hydrazones, arenes, alkenes, and allyl ethers. Comparisons of PMDTA with N,N,N',N'-tetramethylethylenediamine (TMEDA) accompanied by detailed rate and computational studies reveal the importance of the trifunctionality and κ2-κ3 hemilability. Rate studies show exclusively monomer-based reactions of 2-bromooctane, cyclooctene oxide, and dimethylresorcinol. Catalysis with 10 mol % PMDTA shows up to >30-fold accelerations (kcat > 300) with no evidence of inhibition over 10 turnovers. Solid-liquid phase-transfer catalysis (SLPTC) is explored as a means to optimize the catalysis as well as explore the merits of heterogeneous reaction conditions.
Figures





Similar articles
-
Sodium Diisopropylamide: Aggregation, Solvation, and Stability.J Am Chem Soc. 2017 Jun 14;139(23):7921-7930. doi: 10.1021/jacs.7b03061. Epub 2017 May 30. J Am Chem Soc. 2017. PMID: 28557426 Free PMC article.
-
Sodium Diisopropylamide in N,N-Dimethylethylamine: Reactivity, Selectivity, and Synthetic Utility.J Org Chem. 2016 Nov 18;81(22):11312-11315. doi: 10.1021/acs.joc.6b02287. Epub 2016 Nov 2. J Org Chem. 2016. PMID: 27768310 Free PMC article.
-
Sodium Diisopropylamide-Mediated Dehydrohalogenations: Influence of Primary- and Secondary-Shell Solvation.J Org Chem. 2019 Sep 6;84(17):10860-10869. doi: 10.1021/acs.joc.9b01428. Epub 2019 Aug 22. J Org Chem. 2019. PMID: 31436099 Free PMC article.
-
Sodium Alkyl(trimethylsilyl)amides: Substituent- and Solvent-dependent Solution Structures and Reactivities.J Am Chem Soc. 2024 Nov 6;146(44):30397-30421. doi: 10.1021/jacs.4c10836. Epub 2024 Oct 24. J Am Chem Soc. 2024. PMID: 39447193
-
Lithium diisopropylamide: solution kinetics and implications for organic synthesis.Angew Chem Int Ed Engl. 2007;46(17):3002-17. doi: 10.1002/anie.200603038. Angew Chem Int Ed Engl. 2007. PMID: 17387670 Review.
Cited by
-
Continuous Flow Preparation of Benzylic Sodium Organometallics.Angew Chem Int Ed Engl. 2022 Jul 25;61(30):e202203807. doi: 10.1002/anie.202203807. Epub 2022 May 11. Angew Chem Int Ed Engl. 2022. PMID: 35416397 Free PMC article.
-
Li vs Na: Divergent Reaction Patterns between Organolithium and Organosodium Complexes and Ligand-Catalyzed Ketone/Aldehyde Methylenation.J Am Chem Soc. 2023 Mar 22;145(11):6562-6576. doi: 10.1021/jacs.3c01033. Epub 2023 Mar 8. J Am Chem Soc. 2023. PMID: 36890641 Free PMC article.
-
Enhancing Metalating Efficiency of the Sodium Amide NaTMP in Arene Borylation Applications.Angew Chem Int Ed Engl. 2022 Jun 27;61(26):e202204262. doi: 10.1002/anie.202204262. Epub 2022 May 5. Angew Chem Int Ed Engl. 2022. PMID: 35420221 Free PMC article.
-
The Versatile and Strategic O-Carbamate Directed Metalation Group in the Synthesis of Aromatic Molecules: An Update.Chem Rev. 2024 Jun 26;124(12):7731-7828. doi: 10.1021/acs.chemrev.3c00923. Epub 2024 Jun 12. Chem Rev. 2024. PMID: 38864673 Free PMC article. Review.
-
Harnessing Organopotassium Reagents for Cross-Coupling with YPhos-Pd Catalysts: Opportunities, Applications, and Challenges.J Am Chem Soc. 2025 Feb 12;147(6):5417-5425. doi: 10.1021/jacs.4c18073. Epub 2025 Feb 2. J Am Chem Soc. 2025. PMID: 39893653 Free PMC article.
References
-
- Schorigin P Synthesen mittels Natrium und Halogenalkylen Ber. 1908, 41, 2711.
-
- Gilman H; Wright GF The Mechanism of the Wurtz—Fittig Reaction. The Direct Preparation of an Organosodium (Potassium) Compound from an RX Compound. J. Am. Chem. Soc 1933, 55, 2893.
- Gilman H; Breuer F Relative Aromaticities. IV. Furyl-Alkali Compounds. J. Am. Chem. Soc 1934, 56, 1123.
-
- Morton AA The Chemical Behavior of the Organoalkali Compounds. Chem. Rev 1944, 44, 1.
-
- Lochmann L; Janata M 50 Years of Superbases Made from Organolithium Compounds and Heavier Alkali Metal Alkoxides. Eur. J. Chem 2014, 12, 537.
-
- Barr D; Dawson AJ; Wakefield BJ A Simple, High-Yielding Preparation of Sodium Diisopropylamide and Other Sodium Dialkylamides. J. Chem. Soc., Chem. Commun 1992, 204.
- Andrews PC; Barnett NDR; Mulvey RE; Clegg W; O'Neil PA; Barr D; Cowton L; Dawson AJ; Wakefield BJ X-ray Crystallographic Studies and Comparative Reactivity Studies of a Sodium Diisopropylamide (NDA) Complex and Related Hindered Amides. J. Organomet. Chem 1996, 518, 85.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

